Applying Disparate Data Sources and Methods for Evaluating Real-World Evidence
Anna Hung, PharmD, PhD, MS, Duke University School of Medicine, Durham, NC, USA
Real-world evidence (RWE) is important for helping to inform clinical guidelines and policies. Keeping on top of the most recent data sources and methods used to generate RWE is helpful for all of us—researchers, clinicians, policy makers, and patients—to ensure we understand the strengths and limitations of various sources of RWE.
In this on-demand session, 4 studies were showcased, demonstrating diversity in RWE data and methods.
How can data from a national electronic health record system be used to monitor policy decisions?
In 2018, sofosbuvir/velpatasvir was recommended for subsidy in Singapore for the treatment of chronic hepatitis C. To evaluate the impact of this subsidy listing, Xue Shi Luo, MS, BSc (Ministry of Health, Singapore) and colleagues conducted a single-cohort prospective study using Singapore’s national electronic health record data to evaluate a wide variety of outcomes, including drug utilization trends (Figure 1), prescribing patterns, and clinical outcomes (in terms of both effectiveness [sustained virological response rate 12 weeks post-treatment] and safety [adverse events]). The researchers then went one-step further to use these data to inform a model to project longer-term impact on outcomes (eg, progression to decompensated cirrhosis, hepatocellular carcinoma, and hepatitis C-associated deaths) and costs. After examining all of these outcomes, Luo concluded that, “All of these findings have justified the adoption and subsidy listing of sofosbuvir/velpatasvir in Singapore’s public healthcare system.”
Figure 1. Utilization trends in the use of sofosbuvir/velpatasvir.
Why add a physician survey to a medical chart review?
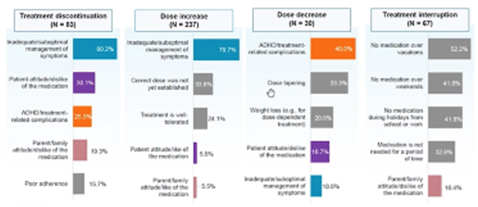
Jeff Schein, DrPH, MPH (Otsuka Pharmaceutical Companies, USA) and colleagues conducted a study related to attention-deficit/hyperactivity disorder treatment that consisted of a chart review and survey of the physicians conducting the chart abstraction. Identified through an online survey panel, 156 physicians abstracted reasons for treatment discontinuation and then were asked to give their perspective on treatment adherence. The chart review revealed common reasons for treatment discontinuation (eg, inadequate/suboptimal symptom management) (Figure 2), while the survey captured information not in the medical charts—such as how often physicians thought their patients’ skipped doses and their subsequent actions (eg, education).
Figure 2. Most common reason for treatment changes.
Both study teams acknowledged that a key limitation of studies relying on electronic health records/medical charts is the exclusion of any information from healthcare services provided outside of the healthcare system. For populations at high risk of dual healthcare system use, this is an important area within RWE that warrants future attention.
What can we do with existing trial data?
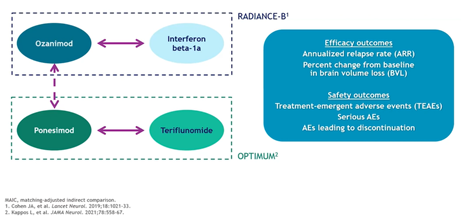
Elyse Swallow, MA, MPP (Analysis Group, USA) discussed their matching-adjusted indirect comparison (MAIC) of trial data to compare 2 medications for the treatment of relapsing forms of multiple sclerosis. Individual-level data was used for one trial while aggregate-level data was used for the second trial, and there was a lack of a common comparator across trials (Figure 3). Swallow also noted that an important challenge to the MAIC method is if there are differences in trial design and inclusion characteristics because such differences cannot be fully adjusted for by the method. However, one key advantage of using clinical trial data is the depth and breadth of treatment-related clinical outcomes, such as relapse rate, brain volume loss, treatment-emergent adverse events, and adverse events leading to discontinuation.
Figure 3. Non-anchor-based matching-adjusted indirect comparison
Does administrative claims data still have a role in RWE?
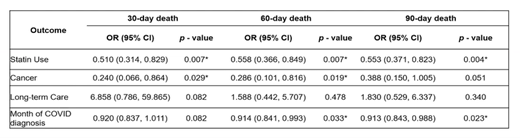
Yiran Rong, MA (PhD candidate at the University of Mississippi, USA) and colleagues conducted a retrospective cohort study applying propensity scores to examine the association between statin use and COVID-related hospitalization and all-cause death in Medicaid-enrolled patients with COVID-19. Study authors found that statin users had lower odds of mortality within 30, 60, and 90 days after diagnosis of COVID-19 (Table 1). One limitation of administrative claims data is the lack of in-depth clinical variables (as compared to electronic health record or trial data), and therefore more narrow scope in outcomes that can be assessed and covariates that are adjusted for. However, because enrollment data are available in administrative claims data, Rong et al were able to exclude those at high risk of dual health insurance use (ie, those dually eligible for Medicare and Medicaid) from their study population.
Table 1. Multivariable analysis between statin users and non-statin users on all-cause death (diagnosed with COVID-19 between March 2020-June 2021)
In summary, there are many advantages and disadvantages to the many different types of RWE. RWE users can gain a better understanding by viewing the full session here: