Generalized Risk-Adjusted Cost-Effectiveness (GRACE): Ensuring Patient-Centered Outcomes in Healthcare Decision Making
Darius N. Lakdawalla, PhD, Schaeffer Center, University of Southern California, Los Angeles, CA, USA; Charles E. Phelps, PhD, University of Rochester, Rochester, NY, USA
SUMMARY
Cost-effectiveness analysis (CEA) is widely used to assess health technology but embeds an assumption at odds with most economic analysis and that conflicts with established understanding of people’s preference patterns. It assumes that health produces happiness with no diminishing returns, in conflict with both standard economic thinking and normal human intuition. Our Generalized Risk-Adjusted Cost-Effectiveness (GRACE) analysis allows diminishing returns to health and aligns CEA with the rest of the health economics literature. This simple change has far-reaching implications for the practice of CEA. In GRACE, optimal cost-effectiveness thresholds are systematically higher for more severe diseases and lower for milder ones than when using standard CEA. We provide formulae for estimating how these thresholds vary with health-related quality of life (QoL) in the sick state. Finally, we provide a coherent way to combine improvements in QoL and life expectancy when people have diminishing returns to QoL. This new approach obviates the need for increasingly prevalent and ad hoc exceptions to CEA for end-of-life care, rare disease, and very severe disease (eg, cancer). Our methods also show that the value of improving QoL for people with disabilities is greater than for comparable people without disabilities, while also incorporating a key patient-preference perspective that QoL gains are more highly valued than life-year extensions for the sickest or most-disabled people. The GRACE model ensures—more than any known system to measure value in healthcare—that patients’ preferences, particularly the quintessential measure of untreated health status, reliably and consistently enter value measures used at both population and individual decision making. This analysis complements and adds to our previous analyses demonstrating how best to incorporate uncertain health benefits into value analyses.
Introduction
Suppose you had psoriasis or acid reflux. Standard cost-effectiveness analysis (CEA) methods would measure your quality of life (QoL) at around 0.9 on a health-index scale from 0 (worst health you can imagine) to 1 (ideal health). How much would you pay for a perfect cure, adding 0.1 extra QoL units? Would it be more than, less than, or the same as what you would pay for 0.1 extra QoL units if you were instead in the latter stages of Duchenne muscular dystrophy (DMD), the ravages of which have left you with 0.25 QoL?
Standard CEA says that improving QoL by 0.1 in each of these very different situations is equivalent. Both intuition and survey data reject this conclusion. Survey respondents regularly state that the same QoL improvement is worth more when given to people in worse health states.1-4 Why does CEA fail to capture this feature of patient preferences, and what does this mean for its other predictions? In particular, the key assumption in standard CEA that leads to the failure to capture the effect of untreated health status also leads directly to 2 other puzzling conclusions. The first is that uncertainty in treatment outcomes does not matter. The second is that people’s willingness to trade life expectancy for QoL improvements (or vice versa) does not vary with baseline QoL.
Standard CEA models decompose health gains into 2 parts: (1) gains in QoL, weighted by remaining (baseline) life expectancy, and (2) gains in life expectancy, weighted by baseline QoL. In this model, a given gain in life expectancy is always worth less to sicker or more-disabled people because life expectancy gains are weighted by lower QoL. Similarly, QoL gains are downgraded if the disability reduces life expectancy.
"Cost-effectiveness analysis has fallen out of step with the rest of health economic research. GRACE aligns the economics of CEA with the human circumstances of patients. It rewards interventions that promote equity and provide relief to patients most in need."
This implication has triggered objections that CEA discriminates against people with disabilities.4,5 Concern about this issue, among others, led to prohibition of using CEA in the United States to measure value in studies by the Patient-Centered Outcomes Research Institute or to determine coverage of medical treatments in Medicare.i
Proponents of CEA say that it has never been used to discriminate against those who are disabled, so there is no problem. However, even absent explicit discrimination, CEA value measurements can influence how healthcare resources are allocated to people with highly severe illnesses and disabilities, how healthcare innovations are reimbursed, and hence, how research and development for new therapies is incentivized.
Several models have been proposed to “fix” this anomaly in CEA. First, the Equal Value of Lives (EVL) method imputes full QoL to disabled people when estimating the value of treatments to resolve their conditions.6 Subsequent research proposed the Healthy Years in Total (HYT) model, which also assumes perfect QoL when valuing gains in life expectancy but adds extra value for gains in QoL itself.5 Both methods address the disability bias issue by assuming ideal QoL when assessing gains in life expectancy. Several European HTA organizations have also considered “proportional shortfall” in health caused by a particular disease as the basis for equity-based weighting of treatment value, but with an ad hoc basis proposed to derive such weights.7
"What’s missing from standard cost-effectiveness analysis? Quite simply, it omits consideration of patients’ starting point—'how sick am I?'”
Within these approaches, analysts must either abandon the rigorous theoretical framework of traditional CEA or employ it while assuming QoL levels that depart from reality. We agree with this issue’s importance, but we believe that these approaches paint over still-unrepaired cracks in the theoretical foundation of CEA.
Instead, we focus on the foundational problem with traditional CEA—its failure to properly reflect how rational consumers would value health improvements in different contexts. Addressing this problem, we analyze how consumers answer fundamental questions about value: (1) “What is my untreated QoL?” and based on that answer, (2) “How much would I value improvements in QoL and additions to life expectancy?” Our model uses a composite measure of health, summarizing multiple dimensions of value into a single composite score (eg, as in EQ-5 health index or comparable multicriteria decision analysis methods).
We use neoclassical economic analysis tools upon which traditional CEA was built, but we eliminate the troublesome and unrealistic assumptions leading to rickety foundations for value assessment. Our analysis reveals how traditional methods fail to account for disease severity, patient risk aversion, and other issues. We prove mathematically that cost-effectiveness thresholds should be higher for more-severe illnesses, and that QoL gains are more valuable to those with worse QoL, whether arising from illness, disability, or combinations thereof. These implications will influence how healthcare resources and innovation investments should be allocated and how medical technology ought to be reimbursed. This analysis extends in new directions our recent study on how uncertainty enters proper value measurement of risk-averse consumers.8
Beyond CEA: Amazing GRACE
Grace: A “polite and thoughtful way of behaving.”
What’s missing from standard CEA? Quite simply, it omits consideration of patients’ starting point—”how sick am I?” People with low QoL will value gains in QoL more than people with greater QoL. Economists call this “diminishing returns.” If you have $100,000 in annual income, $5000 more does not mean as much as when you have $15,000 annual income. If you live in a 5000 square-foot home, adding another 500 square feet is less valuable than if you live in a 1000 square-foot home. In all phases of life, economists observe diminishing returns, formally called “diminishing marginal utility.”
Standard CEA models incorporate diminishing returns in consumption of nonhealth goods and services—things we buy every day. But when it comes to QoL itself, the standard CEA model quite differently assumes that there are no diminishing returns to QoL as it contributes to happiness (utility). This stance is unusual. Indeed, the assumption of diminishing returns to health-related QoL is axiomatic in virtually all of health economics,9-15 except for CEA. We very briefly explore how this assumption distorts the mathematics of cost-effectiveness and then turn to its implications for patient-centered value assessment.
Traditional CEA calculates the incremental cost-effectiveness ratio—added cost (compared to the next-best alternative) divided by added health benefits—and then compares this ratio to a maximum “willingness to pay” cutoff for healthcare, which we will call K.16,17 This willingness-to-pay value is the ratio of income available for consumption, C, to a measure of how quickly diminishing returns set in when consuming nonhealth goods and services. Intuitively, the faster diminishing returns set in on nonhealth consumption, the less “pain” there is in shifting resources to buy medical care. This summary measure of the speed of diminishing returns, ωC, measures the percent gain in utility generated by one percent additional income. If ωC = 1, then there are no diminishing returns, but with diminishing returns, then ωC < 1. Current evidence suggests that 0.3 < ωC < 0.5.16
In the traditional CEA framework, willingness to pay is K = C / ωC . Existing estimates for ωC then imply the willingness-to-pay threshold for CEA is about 2 to 3 times the annual consumption-related income, C. Disease severity and disability do not matter. Once we relax the standard model’s restrictive assumption of nondiminishing returns in QoL, however, wholly different results emerge.
We call our revision to the theory the GRACE model. It changes the willingness-to-pay measure in 2 key ways. First, a new term emerges that accounts for how rapidly returns to health, H, diminish. In parallel to the measure for consumption, we define this speed of diminution as ωH. It has the same meaning as ωC, except that it relates to health-related QoL instead of nonhealth consumption. Second, willingness to pay in GRACE also depends on an index of illness severity (R), which climbs exponentially as baseline QoL degrades. Table 1 provides examples of this pattern for some representative diseases.
Table 1. Example diseases for various levels of l*
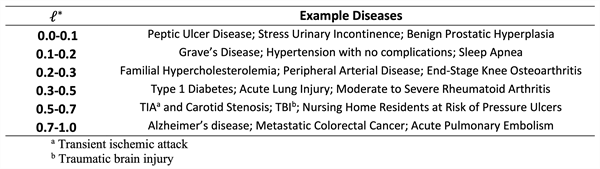
Source: Lakdawalla and Phelps [8]
Quantitatively, R is the ratio of the marginal utility of health in the sick state to the marginal utility when healthy. Combining these 2 changes, the GRACE measure of willingness to pay is not K = C / ωC , but instead is KGRACE = CR [ωH / ωC] = K ωHR. Compared to traditional CEA, GRACE implies lower willingness to pay for treating mild illness (since 0 < ωH < 1 and R ≈ 1 for mild illnesses) but significantly higher willingness to pay for severe illness (since R grows exponentially with illness severity). Traditional CEA causes us to overpay for treatments of mild illnesses but underpay for treatments of severe illnesses.8
By how much are we underpaying for severe illness? The answer to that question depends on how rapidly the incremental value of health changes as the baseline level of health changes. Economists have long understood that the presence and speed of diminishing returns are both linked to a concept called “risk aversion.” Diminishing returns means that 1 unit of consumption—or of health—is worth less to someone who starts with more of it. In turn, this implies that consumers fear the downside of losing 1 unit of consumption more than they value the upside of gaining 1 more unit. Therefore, consumers exhibiting diminishing returns would rather avoid taking risks—even ones whose downside is exactly equal to its upside, like those weighted by the toss of a fair coin. This avoidance of “fair” risky outcomes is the textbook definition of “risk aversion.” In parallel, diminishing returns to QoL imply that patients are averse to taking risks with their health-related QoL. In other words, by ruling out diminishing returns to health-related QoL, traditional CEA assumes—we believe incorrectly—that patients are unconcerned about risks associated with their health and treatment outcomes.
GRACE provides a path forward that accounts for the cost of health-related risks and explains the intuition that disease severity affects value assessment. The standard economist’s measure of “relative risk aversion” over nonhealth consumption is r*C .ii The GRACE model allows for a parallel concept, relative risk aversion in health, denoted as r*H . Standard CEA implicitly assumes that r*H = 0 (ie, patients find it costless to bear risks in their QoL). When we allow instead for values of r*H > 0, the proper way to value gains in health changes dramatically. Now untreated health status—the quintessential patient-centered determinant of value in health—influences willingness to pay for health gains.
To assess the importance of this issue, we need 1 further definition. Again, using QoL scales from 0 (worst health state that you can imagine) to 1 (ideal health), think of a health loss l* measuring the relative change from ideal health. Along this scale, l*= 0.1 is a relatively small loss in QoL, going from ideal health to QoL= 0.9. Alternatively, if l*= 0.5, your health loss would be 50% of the way from “ideal” to “worst imaginable,” and QoL = 0.5. If l*= 0.9, the resulting QoL is 0.1. You would be very, very sick.
Table 1 gives QoL levels for some exemplary diseases or disabling conditions, all drawn from the Tufts University Cost-Effectiveness Analysis Registry (CEAR) data.18 As this table shows, diseases and health conditions that create very large health losses (l* > 0.8) contain many disorders that people think of as very bad health outcomes.
The last piece of the puzzle is the magnitude of r*H . We do not yet have direct measures of this key parameter, but we have good evidence on comparable measures of risk aversion in consumption placing r*C at about 1.0, perhaps a bit lower or higher.19 With no other evidence yet available, we assessed the effects of severity of illness on willingness to pay over a range of values for r*H . If there were a column in Table 2 corresponding to a value of r*H = 0, every entry would be 1.0.
We now turn back to R, the disease severity ratio. As R rises, so does the willingness-to-pay threshold implied by GRACE. The R multiplier depends on relative risk aversion in QoL, r*H ,, and the QoL loss from the disease, l*. Therefore, accurate implementation of the GRACE model will require good estimates of both these parameters. Fortunately, risk aversion over QoL can be estimated “only once” and need not be repeatedly estimated in each specific disease or therapeutic context. It can be estimated with common and well-understood discrete choice experimental methods.20 Moreover, l* is already routinely estimated by “burden of illness” studies and requires no new or specialized estimates. Table 2 illustrates how R varies with both these parameters.
Table 2. R multiplier values in GRACE model for 0 < r*H ≤ 1.3.
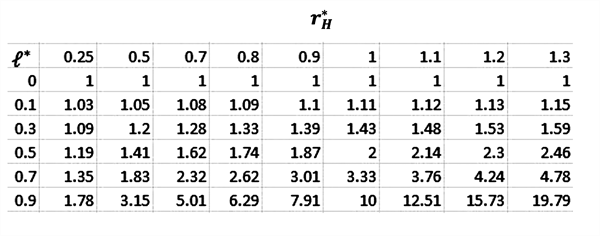
To see how Table 2 works, assume that r*H = 1, similar to central estimates for r*C . Now read down the column under r*H = 1 in Table 2. For low-severity diseases with little QoL loss, R barely exceeds 1.0. As disease severity increases to l* = 0.5 (see Table 1 for exemplary diseases), R = 2, doubling the value per QoL improvement compared to very mild illnesses. As l* reaches 0.7, R grows to 3.33, and for l* = 0.9—very serious conditions—R = 10. Over a reasonably comprehensive range of 0.7 < r*H < 1.3, R varies from 5 to almost 20 for high-severity illnesses. Therefore, the value of improving QoL these conditions (at the margin) is 5 to 20 times larger than for creating similar improvements for low-severity conditions. The same result holds for people with permanent disability—improving their QoL adds great value for severely disabling conditions, in stark contrast to standard CEA measures that lower the value of such improvements.
"GRACE predicts that people would give up more than 3 times the life expectancy in exchange for this quality of life gain, because they place greater value on gains in quality of life."
If the degree of risk aversion in QoL is close to that of risk aversion in consumption, we estimate that the cost-effectiveness threshold for treating mild illness should be around $50,000.8 Table 2 then implies that corresponding thresholds for highly severe illnesses will range from $250,000 up to $1,000,000 per quality-adjusted life-year (QALY) gained. This contrasts with traditional CEA approaches that assume cost-effectiveness thresholds are largely fixed, sometimes with ad hoc exceptions for rare or severe diseases.21
As noted earlier, some European studies propose using proportional shortfall in QALYs caused by a disease as the basis for equity weights to increase imputed value for those in worse health. Our GRACE method provides an economically grounded way of creating the value weights for different disease severity, as distinct from the ad hoc methods suggested in some applications of “proportional shortfall” valuation.
Extensions in Life Expectancy
Acknowledging diminishing returns to and risk aversion over QoL reveals when and how disease severity affects the value of medical technology. GRACE, like virtually all of the standard health economics literature, assumes that people do not have diminishing returns in life expectancy (as opposed to QoL). It also shares this assumption with traditional CEA. Even so, differences in survival valuation still emerge between GRACE and traditional CEA. In the traditional model, the value of extending the probability of survival, p, (ie, life expectancy) is Δp × Baseline QoL. GRACE adds one more term: the value of extending life expectancy must also account for the greater relative willingness to pay for QoL improvements as QoL falls. When people have very low QoL, they are willing to trade more life expectancy to gain improvement in QoL. This is, again, a simple consequence of diminishing returns to QoL.
An example demonstrates this concept using familiar time tradeoff methods. Suppose some people with degenerative knee disease had QoL scores of 0.75, so improvements to full health would add 0.25 to QoL. Suppose these people would give up 0.5 life-years out of 10 remaining expected life-years. Then the tradeoff rate would be 0.25/.05 = 5 additional QoL units demanded per each expected life-year lost.
In contrast, imagine some otherwise-similar people suffering a severe illness such as Alzheimer’s disease or Parkinson’s disease, with a QoL of 0.25. Among this group of people, an improvement to full health would gain 0.75 in QoL score, 3 times the QoL gain as for the “bum knee” patients. However, GRACE predicts that people in this group would give up more than 3 times the life expectancy in exchange for this QoL gain, because they place greater value on gains in QoL. People with greater disability or sickness level are more willing to give up life-years for QoL improvement. This also inherently means they are less willing to give up QoL for life extension.
For given levels of income, people in worse QoL states will have lower willingness to pay for life-years and greater willingness to pay for QoL. Patients’ health context matters when it comes to valuing medical technology. Traditional CEA implies that “a QALY is a QALY is a QALY….” This implication rests on the flawed assumption that the returns to QoL do not diminish. Further, it produces the problematic result that QALY gains are less valuable to sicker or more-disabled people. According to GRACE, the value of health gains to the disabled might be higher or lower: QoL gains are worth more, but the value of pure life expectancy gains remains ambiguous.iii This incorporates patients’ circumstances in ways that traditional CEA methods overlook.
Applications in Decision Making
We see 4 levels of decision making where application of GRACE could move healthcare decisions towards more patient-centered outcomes.
The first step in the decision-making ladder involves health plans’ coverage determinations (those of individual plans or national plans such as the BNHS/NICE in the United Kingdom). For QoL gains, GRACE would reduce values for mild-disease treatments, but greatly increase them for interventions treating high-severity diseases. Full adaptation of GRACE would shift portfolios of covered services towards more-severe diseases, possibly leaving low-severity diseases outside of covered services, as Denmark’s national health plan does now.22 The GRACE model clearly emphasizes that treatments of sicker people have more value, so wider adoption of GRACE would lead to more use of severity-of-illness–based decision thresholds.
Next in the decision ladder comes prior authorization and other formulary restriction rules. Insurance companies do not have different rules for disabled and nondisabled when it comes to applying rules of access to approved treatments, but they regularly exclude coverage of “experimental” treatments, which will most often affect those with the lowest health status. GRACE leads to greater, not reduced access to such interventions for severely sick or disabled persons.
Third, properly constructed decision-support models using the GRACE model could also assist in individual decision making, giving proper “advice” based on patients’ severity of illness (eg, in choosing among alternative cancer therapies).
Finally, as adoption of the GRACE method expands, responding to the shift in reimbursement, research, and development efforts of biotechnology companies and medical centers should shift towards discovery of interventions (drugs, devices, or new procedures) that provide benefit to the most severely ill. As these shifts in research and development occur, new technologies will provide more QoL gains than would occur using current CEA methods, the primary beneficiaries being those with the worst QoL status (pre-existing disease or disability). This approach would also promote equity along the specific dimension of severity of illness because those in the worst health will receive the greatest efforts towards improvement. To the extent that severity of illness is correlated with other measures of inequity, such as socioeconomic class or race, shifting to more generous payments for treatment of severe diseases would also indirectly benefit people along other dimensions of equity.
CEA has fallen out of step with the rest of health economic research. In the process, it has also excised recognition of how patients’ circumstances affect the value of treatment. Health improvements come in an array of types. Consequently, the “QALY is a QALY is a QALY” mantra comes off as especially jarring to real-world patients. It has also long troubled leading practitioners of and advocates for the use of CEA.24,25 In contrast, GRACE aligns the economics of CEA with the human circumstances of patients. It rewards interventions that promote equity and provide relief to patients most in need.
No other measure of patient-centered value measurement exists, we believe, that incorporates the most fundamental questions on the minds of people who are ill or have disabilities: “How sick am I?” and “How much value would I place on improving my QoL?” That, we believe, is the ultimate test of whether a value measurement system is truly patient centered. •
Footnotes
i Specifically, Section 1182, 42 U.S.C. 1320e–1(c(1)) reads: “The Patient-Centered Outcomes Research Institute established under section 1181(b)(1) shall not develop or employ a dollars-per-quality adjusted life-year (or similar measure that discounts the value of a life because of an individual’s disability) as a threshold to establish what type of healthcare is cost-effective or recommended. The Secretary shall not utilize such an adjusted life-year (or such a similar measure) as a threshold to determine coverage, reimbursement, or incentive programs under title XVIII.’’
ii Formally, where U(C) is the utility of a given level of consumption, relative risk aversion in consumption is given by r*C = - CU’’ (C) / (U’ (C) . It measures how fast marginal utility changes as the level of consumption changes.
iii Recent analysis by the authors23 shows how the value of LE gains could either rise or fall as disability increases.
References
1. Nord E, Richardson J, Street A, Kuhse H, Singer P. Maximizing health benefits vs egalitarianism: an Australian survey of health issues. Soc Sci Med. 1995;41(10):1429-1437.
2. Green C, Gerard K. Exploring the social value of health-care interventions: a stated preference discrete choice experiment. Health Econ. 2009;18(8):951-976.
3. Linley WG, Hughes DA. Societal views on NICE, cancer drugs fund, and value based pricing criteria for prioritising medicines: a cross-sectional survey of 4118 adults in Great Britain. Health Economics. 2013;22(8):948-964.
4. Nord E. Severity of illness versus expected benefit in societal evaluation of healthcare interventions. Expert Rev Pharmacoecon Outcomes Res. 2001;1(1):85-92.
5. Basu A, Carlson J, Veenstra D. Health years in total: a new health objective function for cost-effectiveness analysis. Value Health. 2020;23(1):96-103.
6. Nord E, Pinto JL, Richardson J, Menzel P, Ubel P. Incorporating societal concerns for fairness in numerical valuations of health programmes. Health Econ.1999;8(1):25-39.
7. Mott D. Value-based assessment: the case of proportional shortfall. Health Economics Group website. https://blogs.ncl.ac.uk/healtheconomicsgroup/2014/10/17/value-based-assessment-the-case-of-proportional-shortfall/. Accessed June 10, 2021.
8. Lakdawalla DN, Phelps CE. Health technology assessment with risk aversion in health. J Health Econ. 2020;72:1-16.
9. Grossman M. On the concept of health capital and the demand for health. J Political Econ. 1972;80(2):223-255.
10. Muurinen JM. Demand for health: a generalised Grossman model. J Health Econ. 1982;1(1):5-28.
11. Ehrlich I, Chuma H. A model of the demand for longevity and the value of life extension. J Political Econ. 1990;98(4):761-782.
12. Galama T, Kapteyn A. Grossman’s missing health threshold. J Health Econ. 2011;30(5):1044-1056.
13. Arrow K. Welfare analysis of changes in health coinsurance rates. In: The Role of Health Insurance in the Health Services Sector. 1976;NBER:1-34.
14. Cutler DM, McClellan M, Newhouse JP, Remler D. Are medical prices declining? Evidence from heart attack treatments. Quarterly J Econ. 1998;113(4):991-1024.
15. Hall RE, Jones CI. The value of life and the rise in health spending. Quarterly J Econ. 2007;122(1):39-72.
16. Phelps CE. A new method to determine the optimal willingness to pay in cost-effectiveness analysis. Value Health. 2019;22(7):785-791.
17. Garber AM, Phelps CE. Economic foundations of cost-effectiveness analysis. J Health Econ. 1997;16(1):1-31.
18. Tufts Cost-Effectiveness Analysis Registry. https://cevr.tuftsmedicalcenter.org/databases/cea-registry. Accessed August 25, 2020.
19. Chetty R. A new method of estimating risk aversion. Am Econ Review. 2006;96(5):1821-1834.
20. Noussair CN, Trautmann ST, Van de Kuilen G. Higher order risk attitudes, demographics, and financial decisions. Review Econ Studies. 2013;81(1):325-355.
21. Institute for Clinical and Economic Review. 2020-2023 Value Assessment Framework: Response to Public Comments. 2020 [cited 2020 June 22]. https://icer-review.org/wp-content/uploads/2020/01/2020_VAF_Response_to_Comments_013120.pdf. Accessed June 10, 2021.
22. Reckers-Droog VT, van Exel NJA, Brouwer WBF. Looking back and moving forward: on the application of proportional shortfall in healthcare priority setting in the Netherlands. Health Policy. 2018;122(6):621-629.
23. Lakdawalla DN, Phelps CE. A guide to extending and implementing Generalized Risk-Adjusted Cost-Effectiveness (GRACE). Eur J Health Econ. In press.
24. Williams A. Cost-effectiveness analysis: is it ethical? J Med Ethics. 1992;18:7-11.
25. Dolan P, Shaw R, Tsuchiya A, Williams A. QALY maximization and people’s preferences: a methodological review of the literature. Health Econ. 2005;14:197-208.
Explore Related HEOR by Topic

