Background
ISPOR established the Health Science Policy Council in 2004 to advise the Board of Directors on key research issues in the field of health economics and outcomes research. In 2024, the Board of Directors approved a restructuring of the Council and its objectives. These objectives focus on advising on furthering ISPOR’s scientific and policy-related strategy, oversight of the Society’s Task Forces and Special Interest Groups and collaborating with the ISPOR Chief Science Officer on policy responses and special projects.
Mission
The mission of the Health Science Policy Council is to advise the Society on key scientific research and research policy issues in health economics outcomes research (HEOR).
Objectives
- Advise on and assist in furthering ISPOR’s scientific and policy-related strategy, initiatives and collaborations
- Through the subcommittees, oversee Task Forces and SIGs, including recommendations to the Board of Directors for approval of new groups
- Collaborate with the CSO office to lead policy responses and special scientific projects (e.g., the Top 10 Trends report, white papers, and a research spotlight session at major conferences)
- Periodically evaluate the need to engage external experts (e.g., health or regulatory policy experts) for a specific amount of time, as needed
- Produce Annual Plan and twice-yearly progress reports to the Board
Membership and Structure
The Council is composed of up to 3 Past Presidents, 9 invited HEOR experts (members who work in senior positions with 15+ years in the field, have noteworthy publication records, and provide significant service to the Society, with 3 of these slots held by Avedis Donabedian Outcomes Research Lifetime Achievement Award recipients) , 6 mid-career members (members who have 6-14 years of experience in the HEOR field, with 1 of these slots held by a Bernie O’Brien New Investigator Award recipient) and ISPOR’s Chief Science Officer. The Council includes two committees comprised of representatives from ISPOR Consortia and Networks, other ISPOR Councils, and the Value in Health Co-Editors-in-Chief. The Council is led by an executive committee composed of a Chair, Past Chair and Chair-Elect.
Council Committees
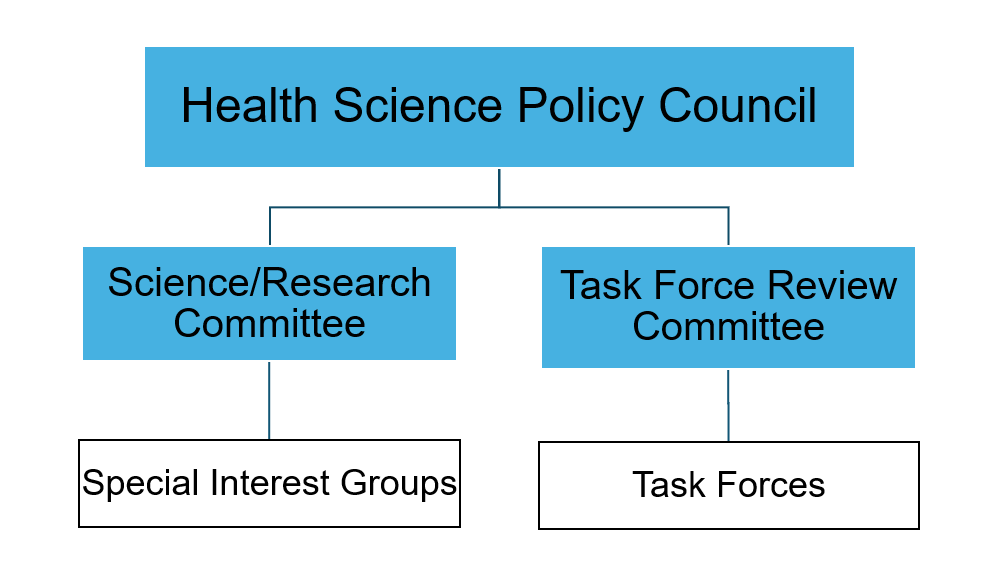
The Health Science Policy Council reports to the ISPOR Board of Directors and is composed of 2 committees.
Health Science Policy Council Chair

Deborah Marshall, PhD, BSc
Health Science Policy Council Committees
Science/Research Committee
- Conducts regular horizon scanning and recommends annual content priorities to the Board of Directors
- Offers input on content for conferences, programs, groups, tools, and other member benefits; input on conference content includes consultation with conference co-chairs during initial conference theme discussions
- Reviews special interest group initial and working group proposals and provides annual review of special interest group activities
- Advises the Board of Directors on, and may support, scientific initiatives and collaborations
Task Force Review Committee
- Reviews initial task force proposals
- Makes recommendation on final task force proposal to the full Health Science Policy Council
- Advises on topics for development
Leadership
Initiatives
The Health Science Policy Council was instrumental in producing the Society's first Top 10 HEOR Trends in 2018 as well as it's successors. The objective of the trends report is to help drive awareness about the important role HEOR plays in informing and improving healthcare decisions.
Council Activities
ISPOR Good Practices for Outcomes Research Task Forces Recommended by HSPC and Approved by the ISPOR Board of Directors
Responses to Calls for Consultation – see Dialogue with Decision-Makers.
