Overcoming Challenges to Innovation in Alzheimer and Parkinson Disease
Alice Beattie, MA; Konstantina Malliou Najjar, MSc; Jasim Uddin, PhD, LCP Health Analytics, London, UK

Alzheimer disease and Parkinson disease are two of the most prevalent and burdensome neurodegenerative conditions worldwide. Alzheimer disease accounts for between 60% and 70% of global dementia cases, while the prevalence of Parkinson disease has doubled over the past 25 years, affecting 8.5 million people in 2019. Both conditions are chronic, progressive, potentially life-limiting, and responsible for a significant and increasing burden on health systems, individuals, carers, and society due to aging populations.
Despite significant investment in neuroscience research, most approved treatments today are limited to symptomatic relief. In recent years, some novel amyloid-targeting, disease-modifying therapies in Alzheimer disease and adjunctive symptomatic treatments in Parkinson disease have received regulatory approval. However, innovation in these areas remains limited, with few disease-modifying options available.
Beyond scientific and regulatory barriers, reimbursement has also emerged as a critical challenge: several approved therapies have secured only limited coverage in the United States, while decisions in the European Union and United Kingdom have been negative or remain pending. Therefore, understanding both the barriers to innovation and the key drivers of reimbursement are essential to identifying targeted actions and engaging healthcare payers in transforming the treatment landscape.
Promises and Pitfalls in Recent Therapeutic Advances
Recent advances in Alzheimer disease have focused primarily on amyloid-targeting therapies. However, access remains limited due to safety concerns and reimbursement challenges. Table 1 summarizes key Alzheimer disease therapies recently appraised by US and European regulatory agencies, together with key issues raised as concerns that later impacted access to the medications.
Table 1. Key Alzheimer Disease Therapies Recently Appraised by US and European Regulatory Agencies

Lecanemab demonstrated clinical benefit with a 27% slowing in cognitive decline over 18 months compared to placebo and was approved by the US Food and Drug Administration (FDA) in 2023, followed by the UK Medicines and Healthcare Products Regulatory Agency (MHRA) in 2024 and the European Commission in 2025.1-3 However, despite the MHRA approval, reimbursement remains a challenge, with the English National Institute for Health and Care Excellence (NICE), in its draft guidance, deciding not to recommend lecanemab due to economic model uncertainties, and negative overall cost-effectiveness estimates.4
Donanemab, another amyloid-targeting therapy, has faced similar challenges. While it was approved by the FDA and MHRA in 2024,5,6 the European Commission initially rejected its marketing authorization application in March 2025 due to the European Medicines Agency’s (EMA’s) safety concerns related to amyloid-related imaging abnormalities; after further assessment, the EMA concluded the benefits of the therapy outweighed the risks, and the European Commission granted marketing authorization in September 2025.7 Both donanemab and lecanemab carry boxed warnings for amyloid-related imaging abnormalities.
Meanwhile, aducanumab received FDA accelerated approval in June 2021, and was subsequently covered in a restricted manner by US public (eg, Centers for Medicare & Medicaid Services [CMS]) and commercial payers, but was still widely criticized by the Institute for Clinical and Economic Review for insufficient evidence of patient benefit and the need for further studies.8 In January 2024, Biogen discontinued aducanumab’s development, reprioritizing their resources towards other Alzheimer disease development projects.9
In Parkinson disease, no therapies have yet been shown to slow disease progression, with recent efforts focused on symptom management. For example, the combination therapy foscarbidopa and foslevodopa, given as a 24-hour subcutaneous infusion, was approved by the FDA in 2024 for advanced Parkinson disease.10 Other recent therapies include opicapone11 and istradefylline,12 which are used as adjuncts to levodopa during “off” periods, when patients experience motor symptoms while taking their usual treatment.
Looking ahead, research and development (R&D) pipelines remain active and contain potential future therapies for both diseases. Table 2 summarizes the current clinical trial landscape in Alzheimer disease and Parkinson disease as of 2024, with 164 active clinical trials investigating drugs for Alzheimer disease, including those targeting prevention and symptomatic relief.13 This included 48 phase 3, 90 phase 2, and 26 phase 1 trials, with 11 long-term extensions. Most (76%) targeted disease modification, while others focused on neuropsychiatric symptom relief (13%) or cognitive enhancement (12%).13 Compared with 2023, there was a slight decline in trials and novel compounds, highlighting ongoing challenges and high failure rates in Alzheimer disease drug development.13
Table 2. Alzheimer Disease and Parkinson Disease Clinical Trial Landscape in 2024
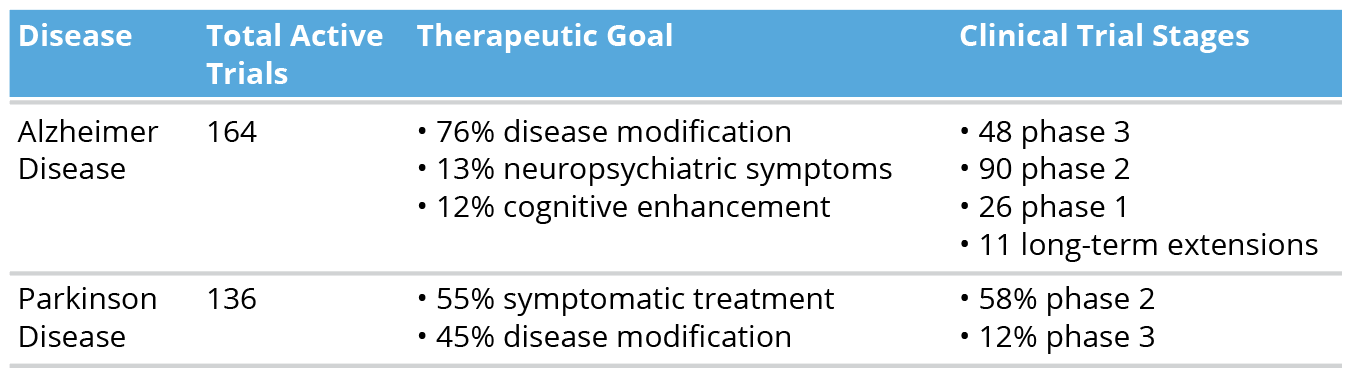
Similarly, as of January 2024, 136 active clinical trials were ongoing in Parkinson disease, with 55% focused on symptomatic treatment and 45% aimed at modifying disease progression.14 Most (58%) were in phase 2, assessing short-term safety and preliminary efficacy, while only 12% had reached phase 3, with just 3 targeting disease modification.14
Despite these active therapeutic pipelines, progress in Alzheimer disease and Parkinson disease is often limited by high attrition and challenges in demonstrating meaningful functional benefits for patients and payers. Tables 3 and 4 illustrate examples of late-stage failures, including reasons for discontinuation across both conditions.
Table 3. Example Late-Stage Failures in Alzheimer Disease Drug Development

Table 4. Example Late-Stage Failures in Parkinson Disease Drug Development

Why Innovation Is Slow in Neurodegenerative Disease
Innovation in neurodegenerative diseases is limited by biological complexity, methodological barriers, and high clinical-trial failure rates. Both Alzheimer disease and Parkinson disease are heterogeneous, slowly progressive conditions, which makes early diagnosis and trial patient selection difficult. Identifying meaningful endpoints is also challenging, particularly in the early stages where symptoms evolve gradually and are hard to quantify. One such example in the case of Alzheimer disease is early episodic memory decline, which evolves slowly and is difficult to capture reliably with current cognitive endpoints. Even when clinical benefits are observed, they may be modest and difficult to translate into functional improvements that resonate with patients and/or payers. For example, modest improvements in Parkinson disease motor scores, particularly those considered to be below or near the minimally clinically important difference, may not translate into functional benefits that meaningfully improve daily life or justify reimbursement.
High R&D development costs and long development timelines further compound these barriers. Demonstrating meaningful impact often requires large-scale, long-duration trials, yet uncertainty around long-term outcomes and limited windows for market exclusivity increase the financial risk for developers. Without clear signals from regulators and reimbursement authorities, manufacturers often hesitate to invest in this space, with market access challenges being a key barrier to innovation.
Market Access Challenges as an Innovation Bottleneck
Even when therapies achieve regulatory approval, securing reimbursement and market access in Alzheimer disease and Parkinson disease remains complex due to clinical uncertainties, economic constraints, and healthcare system capacity limitations. The slow and variable progression of these diseases, combined with limited evidence of sustained functional improvement, makes it difficult to demonstrate value in payer assessments. In addition, the long-time horizon required to observe clinical benefit poses an additional challenge for payers, particularly in systems where patients transition from commercial insurance to public coverage (eg, Medicare), reducing financial incentives for early investment.
Clinical trials often rely on surrogate biomarkers—such as amyloid beta clearance in Alzheimer disease—but their relevance to patient outcomes remains controversial. While some regulators have accepted these endpoints, they are not consistently recognized by health technology assessment bodies and payers. In Parkinson disease, innovation is hampered by the absence of validated biomarkers to assess disease-modifying effects. Even when clinical benefits are observed, they may be modest and difficult to translate into functional improvements that resonate with payers.
In Alzheimer disease, payer skepticism around disease-modifying therapies persists. For example, the CMS requires clinicians to submit data to a CMS-facilitated registry when administering FDA-approved anti-amyloid drugs, as a condition of Medicare coverage.15 Meanwhile, NICE’s draft guidance not to recommend lecanemab highlighted concerns over cost-effectiveness that were partly driven by the uncertainty around long-term benefits.4
Healthcare system readiness and infrastructure requirements (such as diagnostic imaging, infusion delivery, and ongoing monitoring) pose additional challenges, as many healthcare systems are not set up to support the delivery of such services. This lack of preparedness further complicates reimbursement decisions and ultimately hinders the development and adoption of such therapies.
Scientific uncertainty and market access barriers form a reinforcing cycle that continues to slow progress in Alzheimer disease and Parkinson disease, making it difficult for promising therapies to reach patients despite significant research investment.
How Payers Can Incentivize Innovation
To encourage progress in Alzheimer disease and Parkinson disease, healthcare systems should consider how they can play a role in incentivizing development. Traditional quality-adjusted life year-based economic models do not fully capture all possible broader value elements that are particularly important in neurodegenerative disease treatments.16,17 For example, impacts on the wider family, productivity loss, and hospitalizations are all likely to be particularly important for such therapies. Payers should consider how these broader value elements could be integrated into assessments, as they are particularly relevant for aging populations, where societal costs often exceed clinical costs. Additionally, greater alignment of evidence requirements between regulators and payers may also improve access.
In addition to reconsidering how value is measured, innovative pricing models, such as outcome-based agreements and indication-specific pricing, could be considered to align payment with real-world impact. For high-cost therapies with uncertain benefits, installment-based payments or coverage with evidence development can reduce payer risk while maintaining patient access.
Importantly, investment in the disease registries and data platforms that support these flexible reimbursement mechanisms could also support both R&D and postapproval, real-world evidence generation. Additionally, investment in strengthening other key infrastructure such as biomarker testing, imaging, and infusion facilities will be important for enabling timely access.
It is important that governments recognize the importance of innovative strategies to improve reimbursement and access for these therapies. For example, consider higher premiums for disease-modifying therapies than for therapies that provide only symptomatic relief. Lessons from other therapy areas, such as oncology,18 could also be leveraged where initiatives such as the Cancer Drugs Fund in England have helped turn the tide in terms of the lack of innovation beyond chemotherapy.
Payers can play a key role in advancing innovative treatments for Alzheimer disease and Parkinson disease by embracing a mindset that is less skeptical and more strategic. Payer support for broader value assessment frameworks, flexible reimbursement policies, and real-world data repositories is crucial to fully realizing the potential of future therapies for these populations.
References
- McGinley L. Alzheimer’s drug that slows cognitive decline gets FDA approval. The Washington Post. https://www.washingtonpost.com/health/2023/01/06/Alzheimer’s-drug-lecanemab-fda-approval/. Published January 6, 2023. Accessed June 3, 2025.
- Lecanemab licensed for adult patients in the early stages of Alzheimer’s disease [press release]. Medicines and Healthcare Products Regulatory Agency. https://www.gov.uk/government/news/lecanemab-licensed-for-adult-patients-in-the-early-stages-of-Alzheimer’s-disease. Published August 22, 2024. Accessed May 9, 2025.
- European Commission. Commission authorises medicine for treatment of early Alzheimer’s disease [press release]. https://ec.europa.eu/newsroom/sante/items/879055/en. Published April 15, 2025. Accessed March 19, 2026.
- National Institute for Health and Care Excellence. Lecanemab for treating mild cognitive impairment or mild dementia caused by Alzheimer’s disease. https://www.nice.org.uk/guidance/indevelopment/gid-ta11220. Updated January 13, 2026. Accessed March 19, 2026.
- US Food and Drug Administration. FDA approves treatment for adults with Alzheimer’s disease [press release]. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-treatment-adults-Alzheimer’s-disease. Published July 2, 2024. Accessed May 9, 2025.
- Donanemab licensed for early stages of Alzheimer’s disease in adult patients who have one or no copies of apolipoprotein E4 gene [press release]. Medicines and Healthcare Products Regulatory Agency. https://www.gov.uk/government/news/donanemab-licensed-for-early-stages-of-Alzheimer’s-disease-in-adult-patients-who-have-one-or-no-copies-of-apolipoprotein-e4-gene. Published 2024. Accessed May 9, 2025.
- Commission authorises medicine to treat early stages of Alzheimer’s disease [press release]. European Commission. https://health.ec.europa.eu/latest-updates/commission-authorises-medicine-treat-early-stages-Alzheimer’s-disease-2025-09-25_en. Published September 25, 2025. Accessed March 19, 2026.
- US Food and Drug Administration. Aducanumab (marketed as Aduhelm) information. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/aducanumab-marketed-aduhelm-information. Published 2022. Accessed May 9, 2025.
- Biogen to realign resources for Alzheimer’s disease franchise [news release]. Biogen. Published 2024. Accessed May 9, 2025. https://investors.biogen.com/news-releases/news-release-details/biogen-realign-resources-Alzheimer’s-disease-franchise
- US FDA approves VYALEV (foscarbidopa and foslevodopa) for adults living with advanced Parkinson’s disease [press release]. Abbvie. https://news.abbvie.com/2024-10-17-U-S-FDA-Approves-VYALEV-TM-foscarbidopa-and-foslevodopa-for-Adults-Living-with-Advanced-Parkinson’s-Disease. Published October 17, 2024. Accessed March 19, 2026.
- Neurocrine Biosciences announces FDA approval of once-daily ONGENTYS (opicapone) as an add-on treatment for patients with Parkinson’s disease experiencing “off” episodes [news release]. Neurocrine Biosciences. https://neurocrine.gcs-web.com/news-releases/news-release-details/neurocrine-biosciences-announces-fda-approval-once-daily-0. Published April 27, 2020. Accessed March 19, 2026.
- Kyowa Kirin announces FDA approval of NOURIANZ (istradefylline) for use in Parkinson’s disease [press release]. Kyowa Kirin. https://www.kyowakirin.com/media_center/news_releases/2019/pdf/e20190828_01.pdf. Published August 28, 2019. Accessed March 19, 2026.
- Cummings J, Zhou Y, Lee G, Zhong K, Fonseca J, Cheng F. Alzheimer’s disease drug development pipeline: 2024. Alzheimer’s Dement (N Y). 2024;10(2):e12465. doi:10.1002/trc2.12465
- McFarthing K, Buff S, Rafaloff G, et al. Parkinson’s disease drug therapies in the clinical trial pipeline: 2024 update. J Parkinson’s Dis. 2024;14(5):899–912. doi:10.3233/JPD-240083
- Centers for Medicare & Medicaid Services. Monoclonal antibodies directed against amyloid for the treatment of Alzheimer’s disease. https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=305 Published April 7, 2022. Accessed March 19, 2026.
- Park JY, Marcum ZA, Garrison LP. Toward a broader concept of societal value: family spillovers in Alzheimer’s disease. Int J Technol Assess Health Care. 2021;38(1):e7. doi: 10.1017/S0266462321000593.
- Garrison LP, Baumgart M, El-Hayek YH, Holzapfel D, Leibman C. Defining elements of value in Alzheimer’s disease. Value & Outcomes Spotlight. 2021;7(2 S1):S7-S11.
- Willsdon T, Lawlor R, Nicholl H, Douse L. Prioritising Alzheimer’s disease policy in Europe: Insights from oncology. Charles River Associates. https://media.crai.com/wp-content/uploads/2025/05/30123030/CRA-Biogen_Prioritising-Alzheimer’s-Disease-Policy-in-Europe_r_05302025.pdf. Published April 2025. Accessed March 19, 2026.

