From Aspiration to Action: Advancing Patient-Centered Evidence Globally at ISPOR
Clarissa Cooblall, MPH, Senior Director, Scientific Initiatives, ISPOR; Sahar Alam, MPH, Senior Manager, Scientific Initiatives, ISPOR
The landscape of healthcare research is evolving. Patients are not just subjects but active contributors, valued for their lived experiences, perspectives, and expertise. ISPOR—The Professional Society for Health Economics and Outcomes Research—is leading this transformation. It is redefining the role of patients in health economics and outcomes research (HEOR) through an ambitious and inclusive strategy for patient engagement and patient-centered evidence.
ISPOR envisions a global healthcare system where patients are not passive recipients of care but equal partners in shaping the policies, innovations, and research that affect their lives. ISPOR’s longstanding commitment to elevating patient perspectives is based on the belief that meaningful and authentic patient involvement enhances the quality, relevance, and equity of healthcare solutions worldwide.
ISPOR’s commitment to patient-centered evidence is operationalized through its Patient Engagement in HEOR Strategic Initiative (Figure 1), designed to make patient representatives cocreators, not just participants, of healthcare research and decision making.1 This commitment is further underscored in the ISPOR Top 10 HEOR Trends,2 which identifies patient centricity as a priority for 2026–2027. Complementing this, ISPOR’s Good Practices Reports3 demonstrate a sustained focus on patient-centered research, with nearly half of more than 75 published reports dedicated to this area, offering critical methodological guidance and advancing best practices in the field. ISPOR’s HEOR by Topic page on Patient-Centered Research further amplifies this work by providing a centralized and comprehensive platform for knowledge, insights, and best practices.4
Figure 1. ISPOR Patient Engagement in HEOR Initiative
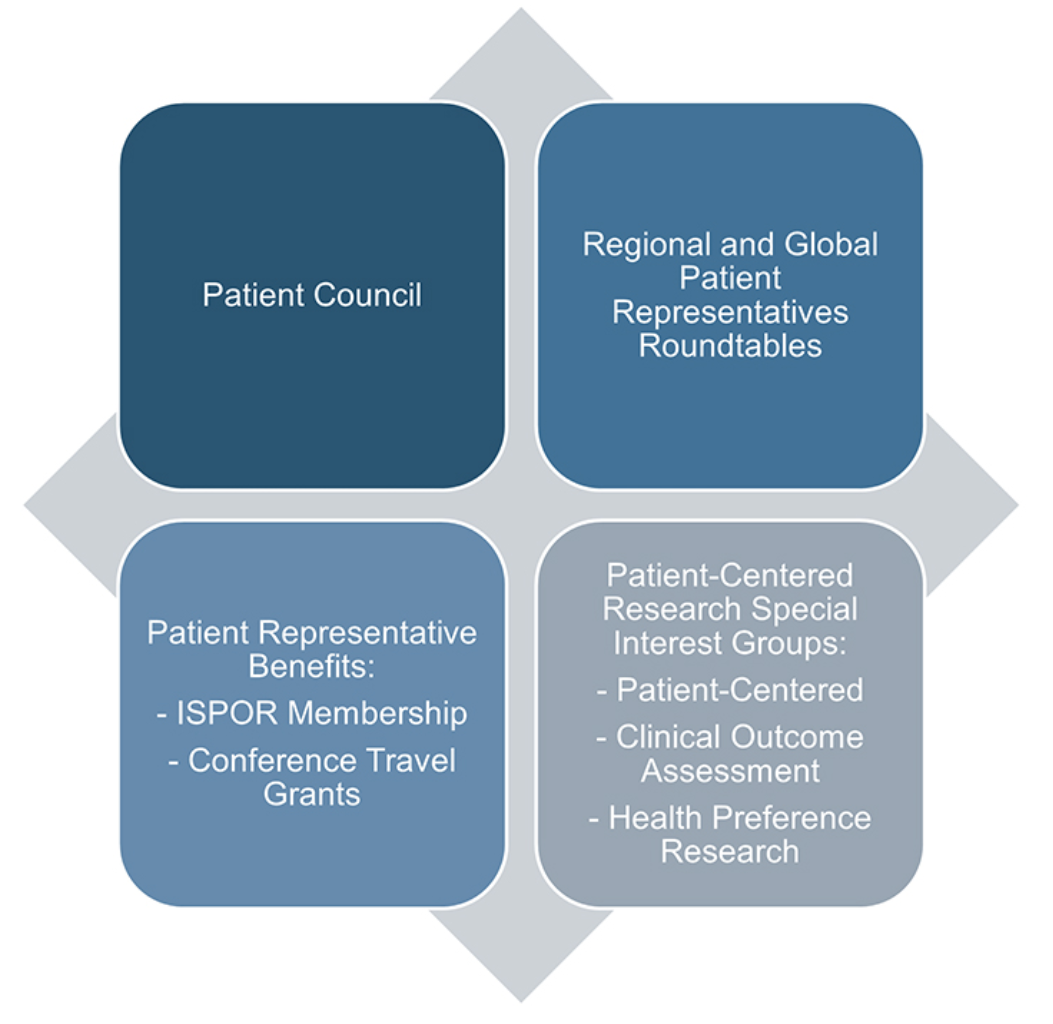
Defining Authentic Patient Engagement
In collaboration with academic and patient advocacy leaders, ISPOR has helped articulate a robust definition of “patient engagement in research”: The active, meaningful, and collaborative interaction between patients and researchers across all stages of the research process, where research decision making is guided by patients’ contributions as partners.5
This definition emphasizes a transition from tokenistic patient involvement to genuine partnership. It is also actively applied across ISPOR’s scientific activities, including conference and Value in Health submissions, where it guides expectations for meaningful patient engagement in research. The ISPOR Patient-Centered Special Interest Group is working to further evolve this definition to incorporate caregiver perspectives, reflecting a broader, more inclusive understanding of lived experience.
Despite growing recognition of its importance, challenges remain. Results from the “ISPOR Membership Survey: Measuring and Evaluating the Quality of Patient Engagement Activities” conducted by this Special Interest Group revealed a significant disconnect: While more than three-quarters of respondents identified their work as “patient-centered,” fewer than half correctly aligned this with involving patients as partners in research, rather than only as study participants. This gap highlights the need for clearer frameworks, education, and standardized practices that promote authentic engagement.6
The ISPOR Patient-Centered Research Summit 2024
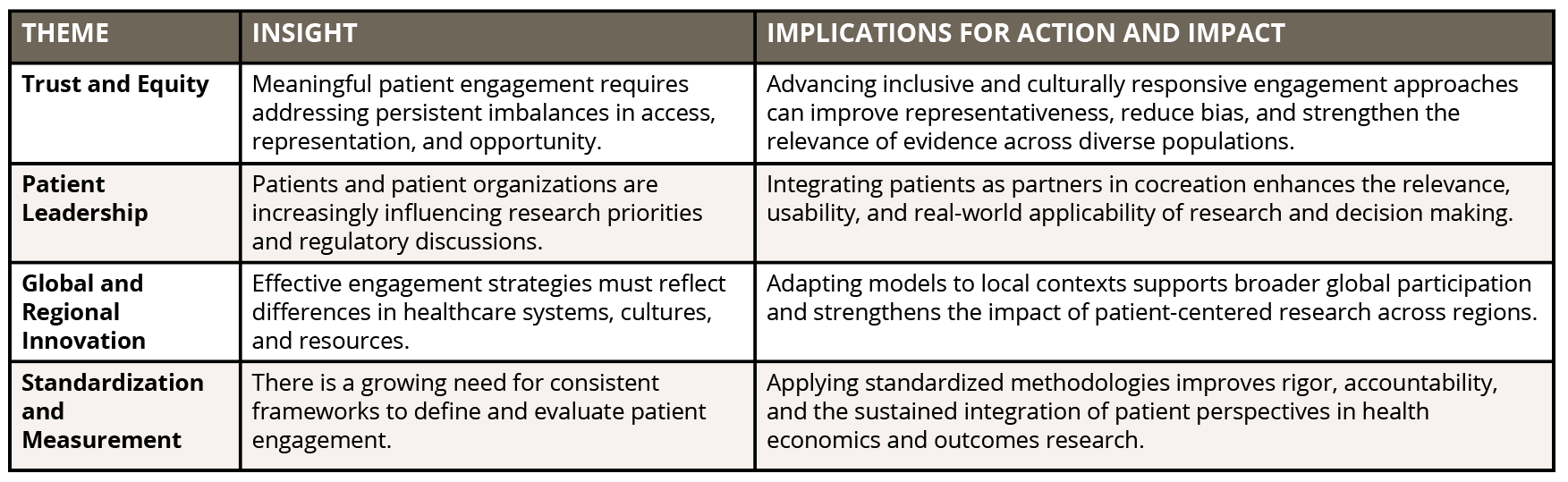
On May 5, 2024, ISPOR hosted its inaugural Patient-Centered Research Summit in Atlanta, Georgia, convening more than 200 participants from more than 20 countries, including patients, researchers, policy makers, industry leaders, and regulatory bodies. The Summit provided a global platform to celebrate progress, share best practices, and cocreate solutions for advancing patient-centered research. The key themes from the Summit are identified in Table 1.
Together, these insights highlight a clear path forward—one in which patient engagement is systematically embedded to strengthen the quality, relevance, and impact of HEOR.
Table 1. Key Themes from the ISPOR 2024 Patient-Centered Research Summit
ISPOR Europe 2025: Patient-Centered Evidence Track
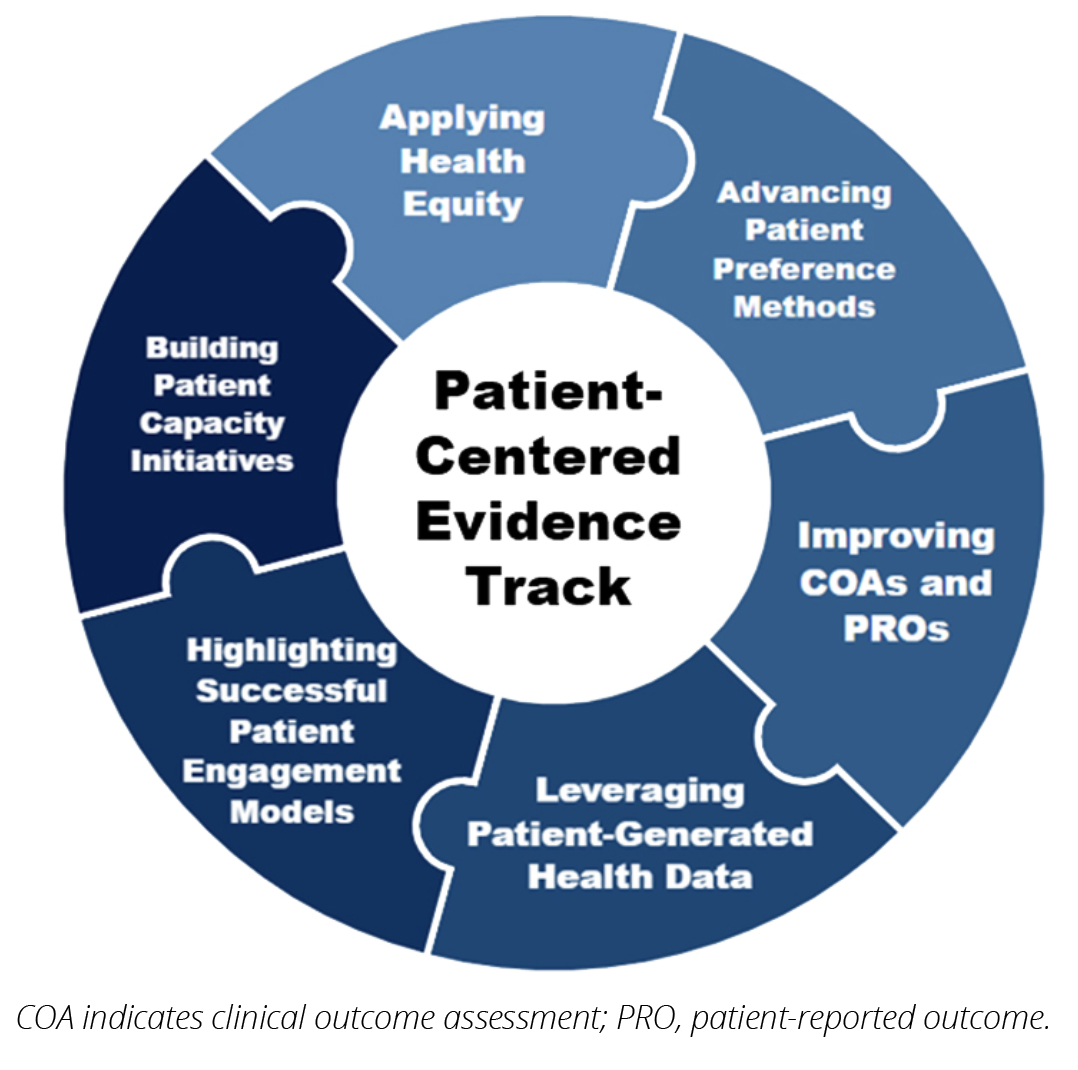
Building on this Summit and integrating the content in the full conference programming, ISPOR launched the Patient-Centered Evidence conference track at ISPOR Europe 2025. The track positioned patient engagement as a scientific standard, showcasing research cocreated with patients and integrating their voices throughout the HEOR process. These sessions recognized patients’ expertise and lived experiences, demonstrating how collaboration enhanced the relevance, usability, and impact of research. Within this conference track, 40% of submitted abstracts were identified as patient-centered. Additionally, 21% of all abstract submissions reported engaging with patients in the research process. The track highlighted cutting-edge research and case studies across several domains, identified in Figure 2.
Figure 2. ISPOR Europe 2025 Patient-Centered Evidence Conference Track Priority Areas
Global Dialogue in Action: The ISPOR Global Patient Representatives Meeting
In January 2026, building on its patient engagement momentum, ISPOR convened 40 global leaders in patient-centered research from across the United States, Canada, Latin America, Europe, Asia-Pacific, and Africa. Discussions centered on embedding patient perspectives more systematically across the evidence ecosystem, with particular focus on the growing importance of patient experience data (PED). Participants examined emerging regulatory developments, including the reflection paper on PED from the European Medicines Agency (EMA), and explored how evolving expectations are shaping opportunities for sustained patient involvement in research, health technology assessment (HTA), and policy. Building on recent methodological advances, regulatory developments from EMA and the US Food and Drug Administration (FDA) emphasizing the importance of incorporating PED, and insights from this meeting, the ISPOR Patient Council will host a session at the upcoming ISPOR 2026 conference on Advancing Patient Experience Data to Strengthen Patient-Centered Real-World Evidence.
Capacity building was a key theme highlighted from participants at the Global Patient Representatives Meeting, with dialogue highlighting tools and strategies to strengthen patient representatives’ roles throughout the research life cycle. The meeting generated actionable insights to inform ISPOR’s evolving patient strategy, reinforced global collaboration, and underscored that lasting progress in HEOR depends on structured partnership and shared accountability.
The Future of Patient Partnership in HEOR
ISPOR continues to advance patient engagement in HEOR by actively gathering perspectives through dedicated events for patient representatives. By convening and engaging in interactive discussions, ISPOR creates opportunities for patients, researchers, and global stakeholders to share insights, highlight priorities, and identify barriers to meaningful participation. These dialogues inform and refine ISPOR’s Patient Engagement in HEOR Strategic Initiative, ensuring that ISPOR’s strategies and programs are grounded in real-world experiences and address the evolving needs of patients worldwide. Through this approach, ISPOR integrates diverse perspectives into its Patient Initiative, shaping a future where patient engagement is embedded at every stage of research and healthcare decision making.
In parallel, ISPOR actively engages with global decision makers and regulatory bodies, including the FDA and EMA, by contributing patient-informed perspectives to consultations and guidance development. This helps ensure that evolving regulatory and policy frameworks reflect real-world patient experience and priorities.7-11
As ISPOR looks to the years ahead, its commitment to elevating patient engagement and partnership is unwavering. A significant step in this direction is the addition of a question on patient engagement in the ISPOR conference submission process—an initiative designed to elevate awareness of genuine patient-centered research and encourage researchers to actively involve patients as partners throughout the research life cycle.
This commitment is further reflected in the breadth of programming at ISPOR 2026, which will feature dedicated forums, such as Collaborating With Patients to Define Digital Endpoints and Biomarkers That Truly Matter and Making Preferences Count: Patient Engaged Values Clarification for Individual-Level Decision Making. Complementing these sessions, ISPOR will offer a short course on Designing a Patient-Centered Strategy for Drug Development and Value, as well as research poster tours focused on PROs and Patient Preference. These efforts are supported by the ISPOR Patient-Centered Research Topic, which highlights patient-centered research sessions across the conference and reinforces a structured approach to embedding patient perspectives in scientific research and evidence.
ISPOR’s global commitment to patient-centered research aligns with the theme of the ISPOR Asia Pacific Summit 2026, “The Future of Health Innovation: Unlocking Affordability and Access.” Sessions will examine the expanding role of real-world evidence, artificial intelligence, and patient-centered research in informing evidence-based decision making, while addressing evolving definitions of value that integrate clinical, economic, and patient-experience perspectives. Individuals with ideas, comments, or an interest in helping define affordability from the patient perspective are encouraged to submit an abstract or reach out via email here.
ISPOR will continue to expand these efforts through initiatives such as the Patient-Centered Evidence conference track at ISPOR Europe 2026, with a focus on patient experience data. This track is dedicated to advancing the science of patient engagement by recognizing and leveraging patients’ expertise, lived experiences, and knowledge to inform evidence generation.
Additionally, ISPOR’s Strategic Plan 2030 focuses on a whole health approach and accounts for how patients define value considering their goals, preferences, and lived experiences. Whole health redefines value by placing patients and their families at the center, ensuring that outcomes, experiences, and priorities that matter most to them drive decision making and innovation.
Furthermore, patient-centered innovation empowers key stakeholders, including investors and early-stage innovators, to systematically engage with patients early. Translating patient insights into decision-relevant learning can inform strategy, strengthen evidence generation, and enhance long-term value creation. More about this topic will be covered at the ISPOR Healthcare Investment Summit 2026.
How to Get Involved
There are multiple avenues for individuals to engage with the ISPOR Patient Engagement in HEOR Strategic Initiative.
- Interested individuals from the global HEOR community are welcome to connect with the ISPOR Patient Council to learn more about ongoing initiatives and opportunities for collaboration and provide ideas and feedback on the Initiative.
- ISPOR also offers a range of engagement platforms, including patient-centered research Special Interest Groups (Clinical Outcome Assessment, Health Preference Research, and Patient-Centered), Task Force Review Groups, and patient-centered research Online Communities.
- In addition, ISPOR supports patient representative participation in its conferences through the ISPOR Patient Representative Travel Grants, with the next application cycle for ISPOR Europe 2026 opening in Summer 2026.
- Email patientcouncil@ispor.org with questions, comments, and ideas.
Together, these opportunities ensure that patient voices are meaningfully represented and integrated across ISPOR’s scientific and collaborative activities.
Helpful Links and Resources:
- HEOR by Topic: Patient-Centered Research
- Special Interest Groups
- Strategic Initiative: Patient Engagement in HEOR
- Good Practices Reports & More
- Upcoming ISPOR Events
Acknowledgements
ISPOR gratefully acknowledges the contributions of many individuals whose expertise, guidance, and dedication have supported ISPOR’s patient engagement initiatives. We extend special thanks to the ISPOR Patient Council, Patient Representatives Roundtable Chairs, leadership teams of ISPOR Patient-Centered Research Special Interest Groups, participants of ISPOR Patient Initiative meetings, and the following individuals: Alan Balch, Bryan Bennett, Angie Botto-van Bemden, Bill Byrom, Mabel Crescioni, Maria Dutarte, Ratna Devi, Pru Etcheverry, Simon Fifer, Ryan Fischer, Laureline Gatellier, Celina Gorre, Sabrina Hanna, Marieke Heisen, Anke-Peggy Holtorf, Francois Houyez, Beyza Klein, Vivian Lee, Maria Mavris, Kenneth Mendez, Derick Mitchell, Marjorie Morrison, Chris Munoz, Axel Mühlbacher, Dionne Ng, Elisabeth Oehrlein, Eleanor Perfetto, Lisa Pieretti, Laura Pizzi, Nan Qiao, Dawn Richards, Kimberly Richardson, Jessica Roydhouse, Katja Rudell, Eva Maria Ruiz de Castilla, Paul Scuffham, Suz Schrandt, Jonathan Stokes, Michael Ward, Durhane Wong-Rieger, and Ziwen (Lily) Zhu.
References
- ISPOR. Patient Engagement in HEOR. https://www.ispor.org/strategic-initiatives/patient-initiatives. Accessed March 18, 2026.
- ISPOR Top 10 HEOR Trends. https://www.ispor.org/heor-resources/top-10-heor-trends Accessed March 18, 2026.
- Harrington, Hanna, Oehrlein, et al. Defining Patient Engagement in Research: Results of a Systematic Review and Analysis: Report of the ISPOR Patient-Centered Special Interest Group, Value Health. 2020;23(6):677-688. https://doi.org/10.1016/j.jval.2020.01.019.
- ISPOR HEOR by Topic: Patient-Centered Research. https://www.ispor.org/heor-resources/heor-by-topic-new/patient-centered-research. Accessed March 18, 2026.
- ISPOR Good Practices Reports & More: Patient-Centered Research. https://www.ispor.org/heor-resources/good-practices/patient-centered-research. Accessed March 18, 2026.
- Llewellyn S, Qiao N, Masurkar P, et al. Quality of Patient Engagement Activities in Health Economics and Outcomes Research: Insights from the ISPOR Community. Value & Outcomes Spotlight. 2024;10(6). https://www.ispor.org/publications/journals/value-outcomes-spotlight/vos-archives/issue/view/value-assessments/quality-of-patient-engagement-activities-in-health-economics-and-outcomes-research--insights-from-the-ispor-community
- ISPOR. Comment submitted to European Medicines Agency on “Reflection paper on patient experience data.” https://www.ispor.org/docs/default-source/strategic-initiatives/ispor-response-to-ema-ped-reflection-paper---final---january-2026.pdf. Published January 30, 2026. Accessed April 17, 2026.
- ISPOR – The Professional Society for Health Economics and Outcomes Research. Comment submitted to US Food and Drug Administration on “Incorporating Voluntary Patient Preference Information Over the Total Product Life Cycle.” Docket (FDA-2015-D-1580-0040). Comment ID #FDA-2015-D-1580-0047. https://www.regulations.gov/comment/FDA-2015-D-1580-0047 Published December 3, 2024. Accessed April 17, 2026.
- ISPOR – The Professional Society for Health Economics and Outcomes Research. Comment submitted to US Food and Drug Administration on ”Patient-Focused Drug Development: Incorporating Clinical Outcome Assessments Into Endpoints for Regulatory Decision-Making.” Docket (FDA-2023-D-0026-0002). Comment ID FDA-2023-D-0026-0049. https://www.regulations.gov/comment/FDA-2023-D-0026-0049 Published July 6, 2023. Accessed April 17, 2026.
- International Society for Pharmacoeconomics and Outcomes Research (ISPOR). Comment submitted to US Food and Drug Administration on “Patient-Focused Drug Development: Selecting, Developing, or Modifying Fit-for-Purpose Clinical Outcome Assessments; Draft Guidance for Industry, Food and Drug Administration Staff, and Other Stakeholders.” Docket (FDA-2022-D-1385-0001). Comment ID FDA-2022-D-1385-0018. https://www.regulations.gov/comment/FDA-2022-D-1385-0018 Published September 27, 2022. Accessed April 17, 2026.
- ISPOR. ISPOR Response to: EUnetHTA on “Practical Guideline Direct and Indirect Comparisons, Patient and Clinical Expert Guidance, and Stakeholder Patient Input Template for Joint Clinical Assessments (JCA).” https://www.ispor.org/docs/default-source/strategic-initiatives/ispor-response-to-eunethta21-jca-patient-engagement-august-2022.pdf. Published August 2022. Accessed April 17, 2026.

