Rethinking the Tuberculosis Response in Times of Change: The Current State of Affairs and Perspectives on the Future
Daniel Mora-Brito, MA, IQVIA, Geneva, Switzerland; Carly Rodríguez, PhD, IQVIA, Durham, NC, United States; Fridah Mwendia, MD, MPH, IQVIA, Nairobi, Kenya

Tuberculosis (TB) is one of the oldest
diseases affecting humankind, accounting for 10.8 million cases and killing
approximately 1.23 million people in 2024.1 While preventable and
curable, TB remains the deadliest infectious disease globally, surpassing
COVID-19- and HIV-related deaths combined. TB disproportionately affects the
most vulnerable and impoverished populations, with an increasingly devastating
impact in patients and communities due to comorbidities, drug resistance,
pandemics, climate change, conflict, migration, and catastrophic out-of-pocket
expenditures for those insufficiently covered by national health schemes.
Notable clinical developments in the last decade have renewed the TB landscape, but more and stronger action is needed to better prevent, diagnose, treat, and support people living with TB. In parallel, as deep changes are presently taking place in the global health architecture, drastically reshaping governance, funding and priorities, it is pertinent to review the state of the art of the TB response, reflecting on the most salient issues directly influencing the disease’s trajectory and the last-mile work of civil society and community-based actors.
The Current Pipeline of Medicines and Vaccines for Tuberculosis
In terms of clinical development over
the last decade, the optimization of rifamycins in drug-sensitive regimens is
promising to shorten the treatment of TB from 6 to 4 months. For drug-resistant
strains, after a 40-year stalemate in drug development, bedaquiline was
approved by the US Food and Drug Administration in 2012,2 followed
by several other new compounds such as delamanid and pretomanid in combination
with linezolid and bedaquiline. For patients who previously would have endured
up to 2 years of treatment, these new medicines have led to the recommendation
of several all-oral, shorter 6- to 9-month regimens.3 Many of these
treatments are composed of 3 or 4 drugs, markedly reducing the potential for
toxicity and drug-drug interactions and making treatment more tolerable for
patients. Likewise, advancements for latent TB infection have broadened options
to shorten treatment from upwards of 9 months of daily medications to 3 months
of weekly treatment.4 Six new immunization products in the late
phase of development could replace the Bacille Calmette-Guérin (BCG) vaccine,
which provides insufficient protection and has been the only TB vaccine
available for almost a century.5,6
Despite consequential discoveries in
recent years, the microbiological features of Mycobacterium tuberculosis,
the pathogen responsible for the disease, continue to pose challenges to drug
and vaccine development. This bacillus can evade the host’s innate immunity,
impede drug penetration due to its waxy cell walls, and prolong the amount of
time required for drug efficacy and resistance testing due to its slow growth. Mycobacterium
tuberculosis can also mutate and develop drug-resistant strains, which can extend
treatment.
These steps forward are reassuring, but they are not entirely devoid of obstacles. Experience on the ground reveals several barriers to TB research and implementation:
- Despite the promising efficacy of bedaquiline-containing regimens, their high cost continues to limit widespread use, particularly in low-income settings. Recent access negotiations have been fast-tracking generic alternatives since late 2024, with halved prices in some countries. In the specific case of South Africa, a price reduction from approximately $306 to $173 per course for generics was the result of withdrawing secondary patents and local licensing agreements.7-9
- There is a need to expand clinical trial infrastructure in sub-Saharan Africa to propel large-scale TB studies, especially in settings of high HIV co-infection. New TB vaccine candidates exist, including MTBVAC and VPM1002 in phase III trials.10 Unlike the BCG vaccine, which offers variable and time-limited protection largely confined to childhood, new TB vaccine candidates aim to deliver durable immunity in adolescents and adults to prevent progression to active disease, addressing key gaps in transmission control.
- Current diagnostic tools still have limitations in rural and high-burden settings. Interferon-gamma release assay, molecular bacterial load assay-based diagnostics, and nucleic acid amplification tests, which are used to detect TB, can be very helpful but require further validation and/or investments to enable optimal use.
- Multidrug-resistant tuberculosis remains a significant challenge, requiring complex treatment regimens that pose adherence issues.11 Ensuring timely diagnosis and treatment initiation, as well as patient care throughout treatment, remains pivotal.
Overall, continued investment to develop shorter and safer treatment regimens, alongside immunization tools, is necessary to keep the momentum and the TB response on track. It is equally vital to prioritize financial commitments for implementation science to assess outcomes in global TB programs for active case finding, diagnosis, and treatment of all forms of TB.
Six new immunization products in the late phase of development could replace the Bacille Calmette-Guérin vaccine, which provides insufficient protection and has been the only TB vaccine available for almost a century.
Existing and Emerging Challenges
Fighting and eventually eradicating TB
requires continued efforts by a wide range of stakeholders, whose engagement is
critical to achieving sustained financing; generating new therapies,
diagnostics, and prevention tools; and supporting adherence and awareness. As
with many public health threats, interventions must connect these different
areas to ensure a proper multi-dimensional response.
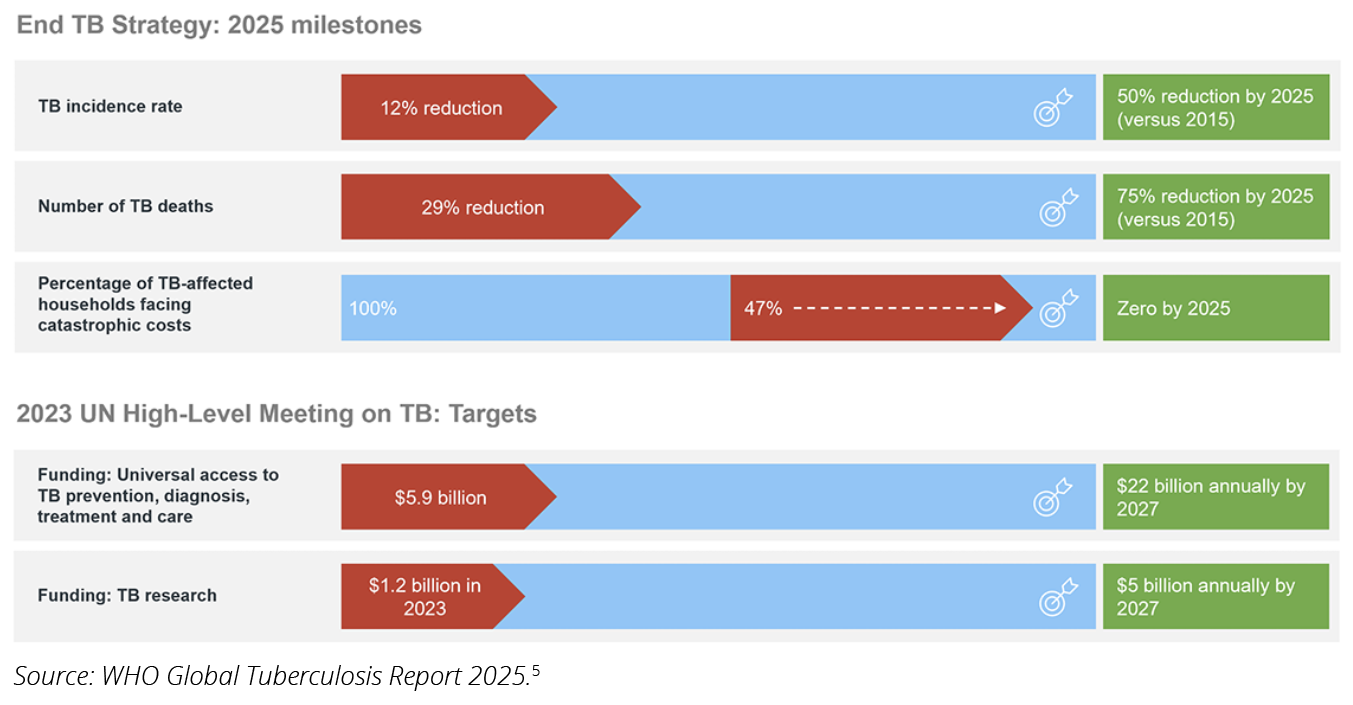
Over the years, many advocacy organizations have signaled the insufficient attention and political commitment that TB has received across a number of dimensions. As illustrated in Figure 1, progress toward the global milestones of the End TB Strategy12 and the targets set by the 2023 UN High-Level Meeting on TB13 has not been small, but considerable gaps are still visible. Although incidence rates were meant to drop around 50% by 2025 compared with 2015, this reduction has not exceeded 12%. Also using 2015 as a baseline, TB deaths have only declined by 29%, when the 2025 target was 75%. On the funding side, the mobilization of $22 billion annually to ensure universal access to TB prevention, diagnosis, treatment, and care has a deficit of approximately $16.1 billion. Resources for research, expected to reach $5 billion annually by 2027 as per the 2025 Global Tuberculosis Report,5 are falling short by 76%. At this stage, 47% of the population living with TB is incurring catastrophic care costs, exposing extensive disparities in universal health coverage.
Figure 1. 2025 Milestones of the End TB Strategy and Targets Stemming from the 2023 UN High-Level Meeting on TB
In an interview conducted for this
article, Ingrid Schoeman, a survivor of extensively drug-resistant TB and
Director of TB Proof, a South Africa-based civil society organization,
underlined the negative effects that contractions in global health funding are
having on TB care. She emphasized the key role that community health workers
play in service delivery, but also in increasing awareness around a disease
whose public perceptions are heavily shaped by profound stigma, and even more
so when coinfections are part of the picture.
“In South Africa, you have people who
concluded their treatment over a decade ago and are TB-free but still are not
invited to family dinners,” Schoeman said.
In this context, according to
Schoeman, more and better awareness and counseling are essential to close
implementation gaps, remove access barriers, and improve care-seeking
behaviors.
“It is important to identify people
who are not accessing TB care, especially within high-risk groups,” she added.
“People who had TB in the past two years, close TB contacts, and those living
with HIV should all be tested regardless of having symptoms. Therefore,
community-led initiatives to raise awareness are extremely important and can
encourage high-risk groups and men, who are the most affected population, to
seek care.”
Schoeman also stressed the importance of factoring in complementary interventions to treatment, including counseling and nutritional needs.
“In South Africa, you have people who concluded their treatment over a decade ago and are TB-free but still are not invited to family dinners.”— Ingrid Schoeman
The Road Ahead: Practical Recommendations to Step Up Action
A push to maintain the cadence and
strength of TB service delivery and related clinical research is urgently
needed. Given the slow progress towards realizing international TB targets,
taking the foot off the gas pedal may jeopardize years of investment and major
achievements in disease control.
The first and most important
recommendation is for every stakeholder in the space to leverage partnerships
and policy advocacy initiatives to accelerate TB elimination. Using an
“all-hands-on-deck” approach, policy makers, research institutions, multilateral
bodies, industry actors, and civil society and community organizations should
find innovative ways to collaborate and tailor creative solutions.
On the research front, it is imperative to:
- Conduct clinical trials and generate real-world evidence for optimal TB diagnostics, vaccines, and treatment in countries and communities that are most affected. Maintaining sufficient investment in the full spectrum of research from phase I to implementation science and service delivery diminishes the likelihood of research pipeline gaps.
- Facilitate cost-effectiveness and modeling studies to drive policy changes in TB drug pricing. A recent study identified that treatment with improved TB regimens of shorter duration may yield substantial cost savings, despite relatively higher drug costs of improved TB regimens compared with the standard of care.14 Moreover, another study estimates that the introduction of a TB vaccine covering adolescents and adults could yield economic benefits in the order of $283 to $474 billion by 2050. Effective prevention tools could be, in this regard, a real game-changer.15
- Strengthen clinical trial networks in Africa to enhance local research capabilities, thereby capturing the manifold genealogical, social, and comorbid conditions of the continent. Research shows that only 4% of the total global clinical trials carried out in 2023 took place in Africa, which results in less than 2% of the data analyzed in genomics research originating from the continent.16 This is concerning when considering that 25% of people who developed TB in 2024 are in Africa, with the Democratic Republic of Congo and Nigeria accounting for 8.7% of global incidences.5 Better and more effective therapies and vaccines need population genomic insight, especially in geographic areas where the disease is more prevalent.
Progress toward global TB milestones and targets has not been small, but considerable gaps are still visible.
On the frontlines of the TB response, nonprofit actors, especially civil society, are increasingly:
- Urging governments to increase domestic resource mobilization, stressing that the benefits of TB prevention and care extend beyond the health sector.17 National health budgets vary broadly, and guaranteeing proper funding for TB care in high-burden countries requires more than a political push for increased allocations. In countries like Brazil, the Russian Federation, India, China, and South Africa, more than 90% of TB financing stems from domestic resources. In other high-burden countries outside this group, dependence on domestic funding can range between 39% and 48%.5 The goal is to increase or maintain resource flows and maximize efficiency gains for national health systems through integrated care approaches.
- Considering alternative and diversified sources of funding, including foundations, multilateral development banks, emerging donors, and the private sector, bearing in mind that these stakeholders cannot entirely or immediately replace the sheer magnitude of public resources traditionally provided to global health. The need to explore alternative sources may invite stakeholders on the ground to think differently and examine interdependencies with other priority areas, but that should not happen at the expense of the values and priorities of key TB populations.
- Leading and fostering partnership opportunities with the private sector to explore innovative solutions, the deployment of blended finance mechanisms, and the design and implementation of access projects—initiatives that will ultimately drive long-term, sustainable solutions.
- Positioning the mandate of relevant TB organizations in new national, regional, or international forums, showcasing how their expertise, networks, and capabilities can impact the TB response.
In light of ongoing challenges and emerging innovations, the global TB response stands at a pivotal juncture. While recent scientific advancements offer unprecedented opportunities to revolutionize prevention and care, structural and financial obstacles continue to hinder progress, particularly in the world’s most affected communities.
It is essential that stakeholders across sectors come together to scale up investments, amplify community-led initiatives, and expand research and development, with an emphasis on high-burden regions. Sustained commitment, bold partnerships, and inclusive strategies will be vital to turning scientific promise into real-world impact and finally closing the equity gap that has long defined the TB epidemic.
References
- World Health Organization. Tuberculosis. https://www.who.int/news-room/fact-sheets/detail/tuberculosis. Published November 13, 2025. Accessed November 17, 2025.
- Mahajan R. Bedaquiline: First FDA-approved tuberculosis drug in 40 years. Int J Appl Basic Med Res. 2013;3(1):1-2. doi: 10.4103/2229-516X.112228
- World Health Organization. WHO consolidated guidelines on tuberculosis. Module 4: Treatment of drug-susceptible tuberculosis. Geneva: World Health Organization, 2022. https://www.who.int/publications/i/item/9789240048126. Accessed March 10, 2025.
- Centers for Disease Control and Prevention. Treatment for latent tuberculosis infection. Published April 17, 2025. https://www.cdc.gov/tb/hcp/treatment/latent-tuberculosis-infection.html. Accessed August 20, 2025.
- World Health Organization. Global Tuberculosis Report 2025. Geneva: World Health Organization, 2025. https://iris.who.int/bitstream/handle/10665/379339/9789240101531-eng.pdf. Accessed December 1, 2025.
- Centers for Disease Control and Prevention. Bacille Calmette-Guérin vaccine for tuberculosis. https://www.cdc.gov/tb/hcp/vaccines/index.html. Published January 31, 2025. Accessed February 20, 2025.
- Dall C. J&J agrees to lower price of TB drug bedaquiline, allow production in South Africa. CIDRAP. https://www.cidrap.umn.edu/tuberculosis/jj-agrees-lower-price-tb-drug-bedaquiline-allow-production-south-africa. Published July 9, 2024. Accessed February 20, 2025.
- Mukwevho N. TB drug price drop is a welcome relief as South Africa faces budget constraints. Health-e News. https://health-e.org.za/2023/11/01/tb-drug-price-drop-is-a-welcome-relief-as-south-africa-faces-budget-constrains. Published November 1, 2023. Accessed February 21, 2025.
- Médecins Sans Frontières. MSF and Health Justice Initiative welcome J&J’s withdrawal of patents on lifesaving TB drug in South Africa. MSF Access Campaign. https://msfaccess.org/msf-and-health-justice-initiative-welcome-jjs-withdrawal-patents-lifesaving-tb-drug-south-africa. Published July 5, 2025. Accessed July 16, 2025.
- Vasiliu A et al. Tuberculosis prevention: Current strategies and future directions. Clin Microbiol Infect. 2024; 30(9):1123-1130. doi: 10.1016/j.cmi.2023.10.023.
- Law S, Daftary A, O’Donnell M, et al. Interventions to improve retention-in-care and treatment adherence among patients with drug-resistant tuberculosis: A systematic review. Eur Respir J. 2019;53(1):1801030. doi: 10.1183/13993003.01030-2018
- World Health Organization. Global Programme on Tuberculosis and Lung Health. https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/the-end-tb-strategy. Accessed March 20, 2025.
- World Health Organization. UN General Assembly high-level meeting on the fight against tuberculosis, 2023. https://www.who.int/activities/preparing-for-the-un-high-level-meeting-on-the-fight-against-tuberculosis--2023. Accessed March 20, 2025.
- Ryckman TS, Schumacher SG, Lienhardt C, et al. Economic implications of novel regimens for tuberculosis treatment in three high-burden countries: A modelling analysis. Lancet Glob Health. 2024:12(6):E995-E1004. doi: 10.1016/S2214-109X(24)00088-3
- Portnoy A, Clark RA, Quaife M, et al. The cost and cost-effectiveness of novel tuberculosis vaccines in low- and middle-income countries: A modeling study. PLoS Med. 2023;20(1):e1004155. doi: 10.1371/journal.pmed.1004155
- Rickwood S, Bailey S, Mora-Brito D. How scaling up clinical research in Africa can benefit society and the economy. World Economic Forum. https://www.weforum.org/stories/2024/08/africa-scaling-up-clinical-research-benefit-society-economy/. Published August 13, 2024. Accessed March 3, 2025.
- TB Alliance. Cycle of poverty. https://www.tballiance.org/why-new-tb-drugs-cycle-poverty/. Accessed January 21, 2026.
- Wells W, Waseem S, Scheening S. The intersection of TB and health financing: defining needs and opportunities. IJTLD Open. 2024;1(9):375-383. doi: 10.5588/ijtldopen.24.0324

