International Reference Pricing Comes to America: The MFN Policies Explained
Sean D. Sullivan, PhD, University of Washington and Curta Inc, Seattle, WA; Jens Grueger, PhD, University of Washington and Curta Inc, Seattle, WA; Kristi Martin, MA, MPA, Camber Collective, Washington, DC

President Donald Trump’s Most-Favored-Nation (MFN) pharmaceutical pricing policy represents one of the most audacious federal initiatives to address prescription drug costs in American history. The policy has evolved into a multifaceted approach combining voluntary manufacturer agreements with 3 distinct regulatory models: GENEROUS for Medicaid, GLOBE for Medicare Part B, and GUARD for Medicare Part D. Together, these agreements and models are part of a strategy to fundamentally reset the prices that Americans pay for prescription medications by tying domestic prices to those paid in other developed nations.
Given the wide-ranging implications of this policy, it will be useful for the health economics and outcomes research (HEOR) community to understand the US government’s rationale for MFN, the proposed implementation models, voluntary direct-to-consumer (DTC) agreements, supply chain implications, implementation and legal challenges, and potential interactions with the provisions of the Inflation Reduction Act (IRA)—as well as implications beyond the United States. Recognizing that the Trump administration drug pricing policies are evolving, the goal of this paper is to provide a general overview of MFN policies in the United States as of the end of 2025.
Policy Rationale and Framework
The Trump administration premises its approach on the fact that the United States, with less than 5% of the world’s population, generates 50% of global pharmaceutical revenues and approximately three-quarters of industry profits. The administration argues that brand drug manufacturers deeply discount products to access foreign markets and subsidize those decreases through much higher prices in the United States. Research supports this assertion,1 showing that list prices for prescription drugs in the United States are on average nearly 3 times as high as those in other developed countries, with prices for branded drugs more than 4 times as high.
On May 12, 2025, President Trump signed an executive order titled “Delivering Most-Favored-Nation Prescription Drug Pricing to American Patients,” formally launching the current iteration of this policy.2 The order directs the US Department of Health and Human Services to establish MFN drug price targets that apply to Medicare and Medicaid programs. When implemented, MFN prices should represent the lowest gross domestic product (GDP) per capita adjusted price in any country that is part of the Organization for Economic Cooperation and Development (OECD) with a GDP per capita of at least 60% of the United States.
List prices for prescription drugs in the United States are on average nearly 3 times as high as those in other developed countries, with prices for branded drugs more than 4 times as high.
The 3 MFN Models: A Seemingly Coordinated Approach
Below, we present the key characteristics, impacted pharmaceutical benefits, and timelines for the 3 MFN models. We further summarize the details in the Table.
A fourth model was announced at the end of December 2025 called BALANCE (Better Approaches to Lifestyle and Nutrition for Comprehensive hEalth), a voluntary model that specifically targets GLP-1 drug coverage for obesity under the Medicare program. The BALANCE model is beyond the scope of this paper; details are available here.
Table. Characteristics of Proposed Most Favored Nation Demonstration Project
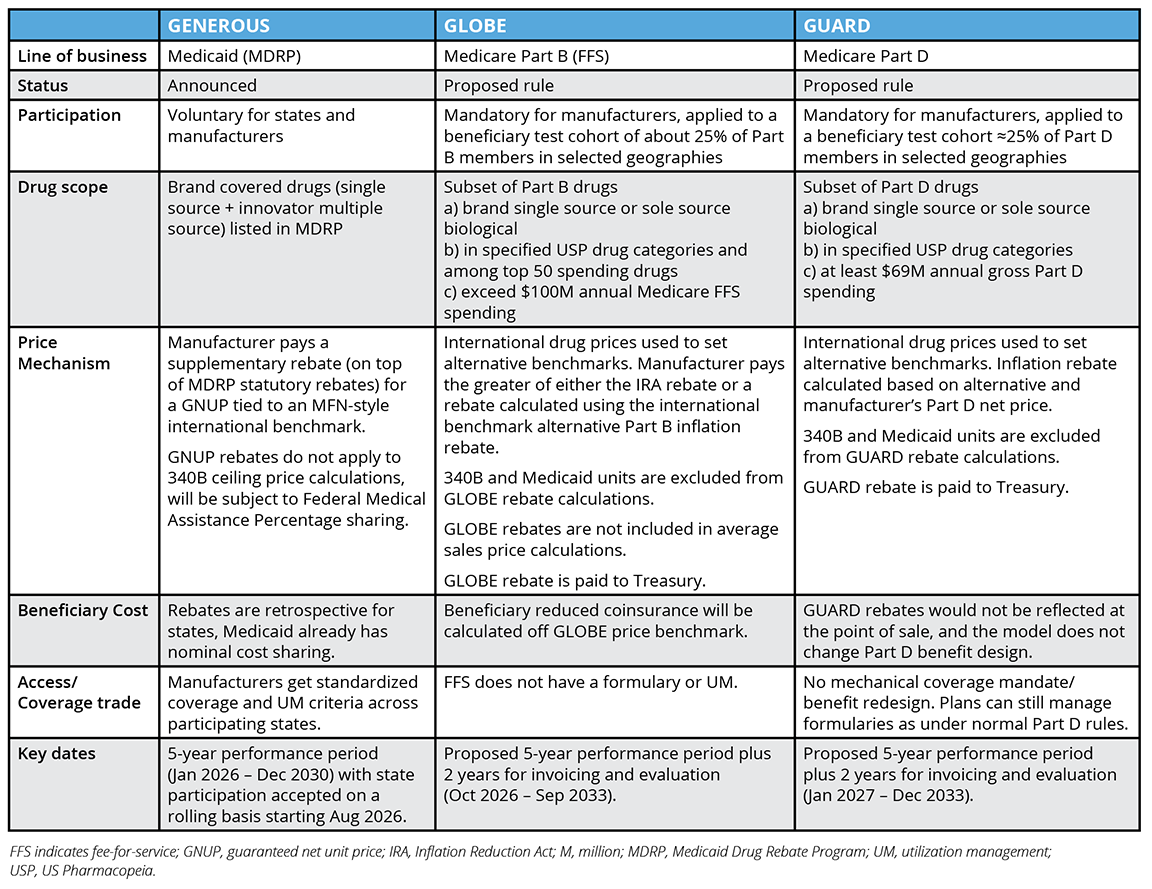
GENEROUS: The Voluntary Medicaid Model
In November 2025, the Centers for Medicare and Medicaid Services (CMS) published the GENEROUS Model (GENErating cost Reductions fOr U.S. Medicaid), a voluntary framework for manufacturers and state Medicaid programs participating in the Medicaid Drug Rebate Program (MDRP).3 GENEROUS relies on voluntary (opt-in) manufacturer and state participation through negotiated supplemental rebates that are in addition to statutory rebates required under the MDRP.
Under GENEROUS, participating manufacturers offer supplemental rebates to participating state Medicaid programs for selected covered outpatient drugs at MFN prices. MFN prices will be identified based on the manufacturer-reported second lowest net price across Canada, Denmark, France, Germany, Italy, Japan, Switzerland, and the United Kingdom, adjusted by gross domestic product using a purchasing power parity method.
The GENEROUS framework creates a marketplace where CMS and manufacturers negotiate using CMS-created standardized coverage criteria and pricing metrics, including utilization management policies and Preferred Drug List placement. Critically, CMS has clarified that supplemental rebates under GENEROUS do not change Medicaid Best Price calculations and therefore do not affect ceiling prices offered under the 340B Drug Pricing Program.4
Manufacturers can submit applications through March 31, 2026, and participation agreements must be executed on or before June 30, 2026. The model includes retroactive pricing to January 1, 2026, and operates without price changes for 5 years, providing states and manufacturers with predictable long-term pricing frameworks.
GLOBE: The (Almost) Mandatory Medicare Part B Model
On December 19, 2025, CMS proposed the mandatory GLOBE model (Global Benchmark for Efficient Drug Pricing),5 designed to incorporate MFN pricing into Medicare Part B. The GLOBE model would test an alternative to the existing Medicare Part B inflation rebates. This proposed model would apply to a subset of the current drugs eligible for inflation rebates, employing a higher rebate for certain high-cost single-source drugs and biological products based on MFN prices, while maintaining some exclusions for drugs such as those purchased under the 340B Program, authorized generics, and drugs with prices in effect under the Medicare Drug Price Negotiation Program (MDPNP).
GLOBE targets provider-administered drugs typically given by infusion or injection in physicians’ offices or hospitals in the outpatient setting. The model applies to 7 therapeutic categories: antigout agents, antineoplastics, blood products and modifiers, central nervous system agents, immunological agents, metabolic bone disease agents, and ophthalmic agents. To qualify, drugs must have Medicare Part B fee-for-service gross annual spending exceeding $100 million and be among the top 50 in spending. CMS provided an illustrative list of 62 drugs based on data from 2024 that may fall under the model based on the criteria as proposed.6
GLOBE (and GUARD, described in the next section) specifies that 19 OECD countries would be used for MFN price benchmarking: Australia, Austria, Belgium, Canada, Czech Republic, Denmark, France, Germany, Ireland, Israel, Italy, Japan, The Netherlands, Norway, South Korea, Spain, Sweden, Switzerland, and the United Kingdom. From that group, CMS will select those countries with purchasing power parity-adjusted GDP per capita meeting specified thresholds, ensuring comparisons are made with economically similar nations.
GLOBE proposes to test 2 international price inflation rebate benchmark methods. Method I would use existing international list price data from publicly available sources like IQVIA MIDAS or NAVLIN, with CMS determining the lowest country-level average list price among reference countries, adjusted for GDP per capita differences. Method II would allow manufacturers to voluntarily submit international net price data on their products. CMS will use the greater of the 2 benchmarks to calculate the rebates owed, adjusted by threshold percentages of 102% for Method I and 105% for Method II, plus incorporating the Medicare Part B add-on payments. This dual-method approach aims to ensure fairness while maintaining administrative feasibility. When prices exceed international benchmarks, manufacturers must pay incremental rebates beyond existing inflation rebates. One substantial issue is the rebate period, which is different in Part B for the IRA rebates (quarter) when compared with the MFN rebates (annual).
For the initial testing phase, the model will operate in randomly selected geographic areas encompassing approximately 25% of Medicare Part B fee-for-service beneficiaries. This geographic limitation allows for controlled testing while minimizing market disruption. Beneficiaries in model-designated areas would see reduced coinsurance, tied to the lower international benchmark rather than the higher US price. The benefit to patients would be similar to the existing coinsurance reduction under the IRA Part B inflation rebates in place today.
GLOBE would start October 1, 2026, and run through September 30, 2031, with rebate invoicing and reconciliation continuing through 2033. CMS suggests that GLOBE will reduce Medicare spending by approximately $12 billion over this period.
To qualify for GLOBE, drugs must have Medicare Part B fee-for-service gross annual spending exceeding $100 million and be among the top 50 in spending.
GUARD: The (Almost) Mandatory Medicare Part D Model
Released simultaneously with the GLOBE proposal, the GUARD model (Guarding U.S. Medicare Against Rising Drug Costs)7 applies similar MFN principles to self-administered drugs covered under Part D and often dispensed at retail pharmacies. GUARD would cover 17 therapeutic classes, excluding drugs with annual spending below $69 million, 340B purchased drugs, and drugs subject to CMS-negotiated maximum fair prices (MFPs) under the MDPNP, avoiding duplication.
GUARD would use the same 19-country reference basket and methodology for determining international list price benchmarks as GLOBE. However, the rebate calculation would differ to reflect Part D’s complex financing and benefit structure. The international benchmark is subtracted from the Medicare “net” price, proposed in the model to be calculated as the drug’s wholesale acquisition cost minus manufacturer rebates to Part D plan sponsors and manufacturer discount program payments. The GUARD rebate is applied when this difference exceeds the inflation rebate that is otherwise required under existing law.
Like GLOBE, GUARD would apply to approximately 25% of Medicare beneficiaries in randomly selected geographic areas. However, unlike GLOBE, GUARD eligibility would include beneficiaries enrolled in standalone Part D plans or Medicare Advantage-Part D plans. GUARD would start January 1, 2027, and run for 5 years with rebate invoicing and payment continuing until 2033. CMS projects that GUARD will reduce Medicare gross spending by approximately $14 billion.
Voluntary Manufacturer Agreements and TrumpRx
Beyond the 3 MFN models, the administration has pursued voluntary manufacturer agreements that have been negotiated directly between the manufacturers and the federal government. In July 2025, President Trump sent letters to the CEOs of 17 pharmaceutical manufacturers with high-priced branded prescription drugs and demanded they lower prices to match those in other developed nations.8 By January 2026, 16 companies had announced agreements9 for selected products: AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Eli Lilly, EMD Serono, Genentech, Gilead Sciences, GSK, Johnson&Johnson, Merck, Novartis, Novo Nordisk, Pfizer, and Sanofi.
The framework of the agreements and specific terms are confidential. Manufacturers typically state in their press releases that they have “committed to launching new medicines with a more balanced pricing approach across developed countries.”
They can choose:
- The products for which they extend these prices to every Medicaid beneficiary
- To contract with the United States to guarantee that Medicare, Medicaid, and commercial payers receive the MFN price for newly launched drugs, and
- To participate in DTC or direct-to-business distribution models. This forward-looking provision aims to prevent future price disparities for newly developed medications, fundamentally changing the pharmaceutical pricing landscape.
In general, the agreements have a duration of 3 years; the relevance of the agreements beyond this period is unknown. A cornerstone of implementation is the TrumpRx platform, a government website that connects patients to purchase medications directly from manufacturers at reduced prices, effectively bypassing health insurance and pharmacy benefit manager intermediaries.
The administration announced that these agreements will deliver dramatic price reductions on high-profile medications.8 NovoNordisk already announced that the prices for the lower dosage forms of Ozempic (semaglutide) and Wegovy (semaglutide) fell from $1000 and $1350 to $349 per month when purchased directly through its DTC platform, NovoCare Pharmacy.10 Sanofi will reduce the price of its blood thinner, Plavix (clopidogrel bisulfate), from $756 to $16 for patients.8 Plavix has been competing against several generic competitors since 2012, so it is unclear whether this newly announced price will make a difference for patients given the current generic price competition.
Investment and Supply Chain Commitments
Beyond pricing commitments, the agreements include language related to domestic manufacturing investments. The pharmaceutical manufacturers involved have committed to investing at least $150 billion collectively in United States manufacturing in the near term. Of note, most of these manufacturing commitments were reached before the announcement of the MFN models. Some CEOs of the involved companies have said publicly that the 2017 federal tax cuts were the catalyst for the domestic manufacturing investments. Companies are also donating active pharmaceutical ingredients to the Strategic Active Pharmaceutical Ingredients Reserve to reduce foreign dependence and ensure adequate emergency supplies, addressing national security concerns exposed during the COVID-19 pandemic.
In exchange for compliance, participating companies receive exemptions from Trump’s planned pharmaceutical-specific tariffs and benefit from priority regulatory review vouchers for new drugs. The agreements provide a 3-year grace period from tariffs, contingent on continued investment in American manufacturing capacity. This carrot-and-stick approach incentivizes both immediate price reductions and long-term domestic production expansion.
Potential Interaction With the Medicare Drug Price Negotiation Program
The relationship between the MFN policies and the MDPNP represents one of the most complex aspects of current pharmaceutical policy. The IRA authorized CMS to negotiate MFPs for certain high-expenditure, single-source drugs without generic or biosimilar competition. Both GLOBE and GUARD explicitly would exclude drugs already subject to negotiated MFPs, suggesting the policies are designed as complementary rather than overlapping mechanisms, which is consistent with the existing Medicare Inflation Rebate program. This strategic separation allows the MFN models to target drugs not covered by Medicare price negotiations, while the negotiation program addresses the highest-spend medications through a more deliberative process.
However, there is potential for conflicts to emerge in implementation. The MDPNP is moving forward under the Trump administration. Meanwhile, voluntary MFN agreements have produced different pricing for the same medications. For instance, the MFP for Novo Nordisk’s semaglutide products is $274 (including Rybelsus), while the voluntary MFN agreement announced a $245 Medicare price (excluding Rybelsus)—creating pricing and coverage ambiguity for identical molecules.
Critics argue that both approaches essentially represent leverage campaigns rather than traditional value-based negotiations. Some pundits characterize the overall MFN strategy as using threats of trade penalties, import restrictions, and regulatory pressure to force price reductions, with the Medicare negotiation authority serving as the domestic counterpart to external MFN pressure on foreign governments. The construction of the MDPNP participation—where manufacturers may face excise taxes if they refuse to participate in negotiations—combined with the coercive elements of MFN implementation suggests these policies work in tandem to maximize federal purchasing power.
The interaction also raises questions about sustainability and future industry innovation. Some policy observers suggest neither approach establishes a principled, predictable pricing framework to reduce prices.11 This could have implications for pharmaceutical research and development investments, although the Congressional Budget Office expects that while the IRA will slow new drug introductions, the effect will be modest.11 The combined pressure from both programs may amplify these concerns.
Looking forward, the third round of Medicare drug price negotiations begins in early 2026, with 15 additional high-cost drugs to be selected. How manufacturers respond to dual pressures from both IRA negotiations and MFN models will likely shape domestic and international pharmaceutical pricing strategy, potentially forcing companies to choose between market access and profit margins in ways unprecedented in American healthcare policy.
A cornerstone of MFN implementation is the TrumpRx platform, a government website that connects patients to purchase medications directly from manufacturers at reduced prices.
Legal Challenges and Implementation Questions
Several manufacturers who have agreed to deals with the Trump administration have reported that their prior MFN deals exempt them from participation in GLOBE and GUARD—thus, our cheeky descriptor “almost mandatory” above. CMS and the administration have not clarified whether such manufacturers are, in fact, exempt. If true, the question remains as to whether the manufacturers are exempt only for specific drugs or their entire portfolio, including future launches. If there were any exemptions included in the MFN agreements, this raises questions about the “mandatory” nature of the models as well as whether the models could be seen as punitive toward manufacturers who did not seek MFN agreements with the administration.
The MFN models may face significant legal hurdles. In 2020, at least 3 federal courts ruled in favor of entities that challenged the first Trump administration’s attempt to implement an MFN-based Medicare Part B model, focusing on Administrative Procedure Act violations, claims of exceeded agency authority, and separation of powers issues.12 This represents Trump’s second and more expansive attempt at MFN pricing, following the 2020 model that was blocked by a federal judge due to procedural failures.
The current approach differs in many ways. First, CMS is following proper notice and comment administrative processes, unlike the previous MFN-based Medicare model that was blocked due to procedural failures in 2020. Second, these models focus on manufacturers, while prior attempts tried to address drug pricing through Medicare provider payment systems. Finally, the GLOBE and GUARD models rely heavily on the IRA and the program operations built under the Medicare Inflation Rebate program authorized under the law, which were not available during the first Trump administration.
Critics contend that drug manufacturers may simply raise list prices in the referenced countries to align with United States prices and that there is no enforcement mechanism for drug manufacturers who do not voluntarily change their prices. This “leveling up” risk could undermine the policy’s effectiveness while potentially harming patient access in reference countries.
Questions also remain about how these models interact with existing programs like the IRA’s Medicare negotiation provisions and the 340B Drug Pricing Program. The complexity of overlapping policies creates implementation challenges and potential unintended consequences for safety-net providers and beneficiaries.
The pharmaceutical industry trade group PhRMA has opposed MFN pricing as the best approach to lowering drug costs, instead pointing to pharmacy benefit managers as contributing to raising prescription drug prices in the United States.13 Despite industry resistance, the policy has garnered some bipartisan interest, with Rep. Ro Khanna (D, California) introducing legislation14 to codify Trump’s executive order, suggesting potential durability beyond the current administration. The Global Fairness in Drug Pricing Act was cosponsored by another Democrat and 2 Republicans.
How manufacturers respond to dual pressures from both IRA negotiations and MFN models will likely shape domestic and international pharmaceutical pricing strategy.
Implications Beyond the United States
A key premise of the United States administration is that lower drug prices in that country will be compensated by higher prices overseas, so revenues for manufacturers will not be materially impacted. In all reference countries, complex price negotiations between manufacturers and national health and statutory insurance systems need to be completed for national reimbursement coverage for new medicines. Inability to achieve a negotiated agreement leads to unavailability of medicines.
Of the 173 new medicines approved by the European Medicines Agency between 2020 and 2023, only 29% are fully available, and 17% have limited availability across 36 European countries.15 While published list prices for these products are typically within a narrow corridor, confidential net price agreements provide flexibility at country level to reflect purchasing power and healthcare priorities. For already marketed products, there are no mechanisms for industry to increase prices.
However, recent pressure from the US Trade Representative in tariff negotiations with selected countries has resulted in some commitments to increase drug budgets and prices. In the United Kingdom, the cost-effectiveness threshold for national reimbursement will be raised by 25% in 2026,16 and drug budgets will be increased.17 Italy has increased drug budgets to allow for higher drug prices.18 Overall, it is unlikely that all reference countries will increase their drug budgets and introduce mechanisms that allow for higher drug prices, and consequently the availability of new medicines will be further reduced, which will have negative implications for manufacturer revenues.
Conclusion
The administration’s layered approach—combining voluntary agreements through GENEROUS with proposed mandatory models through GLOBE and GUARD—represents an unprecedented attempt to implement international reference pricing across the 2 largest payers in the United States simultaneously. Whether this multifaceted strategy will survive legal challenges, achieve sustained price reductions for Americans, and avoid negative consequences for pharmaceutical innovation remains to be seen. Many details of the models remain to be clarified.
The stakes are enormous: Success could save billions in healthcare spending, while failure could entrench existing disparities and undermine future reform efforts. Beyond the United States, MFN may have significant implications for patient access in designated reference countries and beyond.
The consequences for pharmaceutical innovation are difficult to predict, but budgets for research and development will be under pressure as revenue losses for pharmaceutical manufacturers in the United States will not be compensated by higher revenues abroad as the administration has suggested. Fewer new medicines reaching patients in need will negatively affect all countries, including the United States.
While high drug prices are an economic issue for payers, employers, and patients, the United States has been leading in pharmaceutical innovation, providing patients in need with new and more effective treatment options. Outside the United States, access to these medicines is significantly restricted, often because of political cost containment measures that have been heavily criticized by many stakeholders, including policy makers and academics. Rather than importing such cost-containment schemes, the United States needs a more sophisticated system to inform appropriate pricing and coverage of medicines based on clinical, economic, humanistic, and ethical considerations.
References
- Office of the Assistant Secretary for Planning and Evaluation. International Prescription Drug Price Comparisons: Current Empirical Estimates and Comparisons With Previous Studies. United States Department of Health and Human Services. https://aspe.hhs.gov/reports/international-prescription-drug-price-comparisons. Published July 1, 2022. Accessed January 21, 2026.
- The White House. Delivering most-favored-nation prescription drug pricing to American patients. https://www.whitehouse.gov/presidential-actions/2025/05/delivering-most-favored-nation-prescription-drug-pricing-to-american-patients/. Published May 12, 2025. Accessed January 21, 2026.
- Centers for Medicare & Medicaid Services. GENEROUS (GENErating cost Reductions fOr U.S. Medicaid) Model. https://www.cms.gov/priorities/innovation/innovation-models/generous. Updated December 25, 2025. Accessed January 21, 2026.
- Health Resources & Services Administration. 340B Drug Pricing Program. https://www.hrsa.gov/opa. Updated January 2026. Accessed January 21, 2026.
- Centers for Medicare & Medicaid Services. GLOBE (Global Benchmark for Efficient Drug Pricing) Model. https://www.cms.gov/priorities/innovation/innovation-models/globe. Updated December 29, 2025. Accessed January 21, 2026.
- Centers for Medicare & Medicaid Services. Part II.B.6: Illustrative List of Proposed Performance Year 1 Globe Model Drugs and Model Participants. In: GLOBE (Global Benchmark for Efficient Drug Pricing) Model. Fed Regist 2025;90(244):60260-60262. https://www.federalregister.gov/documents/2025/12/23/2025-23702/global-benchmark-for-efficient-drug-pricing-globe-model#h-37. Published December 23, 2025. Accessed January 21, 2026.
- Centers for Medicare & Medicaid Services. GUARD (Guarding US Medicare Against Rising Drug Costs) Model. https://www.cms.gov/priorities/innovation/innovation-models/guard. Updated December 29, 2025. Accessed January 21, 2026.
- The White House. Fact Sheet: President Donald J. Trump announces largest developments to date in bringing most-favored-nation pricing to American patients. https://www.whitehouse.gov/fact-sheets/2025/12/fact-sheet-president-donald-j-trump-announces-largest-developments-to-date-in-bringing-most-favored-nation-pricing-to-american-patients/. Published December 19, 2025. Accessed January 21, 2026.
- Lupkin S. Trump struck deals with 16 drug companies. but they’re still raising prices this year. NPR. https://www.npr.org/2026/01/16/nx-s1-5678915/trumprx-pharma-drug-price-deals-list-prices. Published January 16, 2026. Accessed January 21, 2026.
- Novo Nordisk. Novo Nordisk launches introductory self-pay offer for Wegovy® and Ozempic® for $199 per month [press release]. https://www.novonordisk-us.com/media/news-archive/news-details.html?id=916460. Published November 17, 2025. Accessed January 21, 2026.
- Congressional Budget Office. Estimated Budgetary Effects of Public Law 117-169, to Provide for Reconciliation Pursuant to Title II of S. Con. Res. 14. https://www.cbo.gov/publication/58455. Published September 7, 2022. Accessed January 21, 2026.
- Congressional Research Service. Most-favored-nation prescription drug pricing executive order: Legal issues. https://www.congress.gov/crs_external_products/LSB/PDF/LSB11319/LSB11319.1.pdf. Published June 5, 2025. Accessed January 21, 2026.
- Ubl SJ. ‘Most-favored nation’ policy isn’t the way to lower drug prices. [Originally published by STAT+ on June 26, 2025] https://phrma.org/resources/stat-phrma-ceo-most-favored-nation-policy-isn-t-the-way-to-lower-drug-prices. Published July 1, 2025. Accessed January 21, 2026.
- Reps. Khanna, Luna, Kaptur, and Biggs introduce bill to codify Trump’s executive order to lower prescription drug costs [press release]. https://khanna.house.gov/media/press-releases/release-reps-khanna-luna-kaptur-and-biggs-introduce-bill-codify-trumps. Published May 20, 2025. Accessed January 21, 2026.
- Newton M, Stoddart K, Travaglio M, Troein P. EFPIA Patients W.A.I.T. Indicator 2024 Survey. IQVIA. https://efpia.eu/media/oeganukm/efpia-patients-wait-indicator-2024-final-110425.pdf. Published May 2025. Accessed January 21, 2026.
- National Institute for Health and Care Excellence. Changes to NICE’s cost-effectiveness thresholds confirmed. https://www.nice.org.uk/news/articles/changes-to-nice-s-cost-effectiveness-thresholds-confirmed. Published December 1, 2025. Accessed January 21, 2026.
- Inch A. Changes to medicines policy: what you need to know. NHS Confederation. https://www.nhsconfed.org/publications/changes-medicines-policy-what-you-need-know. Published December 15, 2025. Accessed January 21, 2026.
- Legge di bilancio 2026, le misure per la sanità. Ministero della Salute, Governo Italiano. https://www.salute.gov.it/new/it/news-e-media/notizie/legge-di-bilancio-2026-le-misure-la-sanita/. Published January 2, 2026. Accessed January 21, 2026.

