In Sync With Technological Innovation and Ongoing Evidence Generation: The Need for an HTA Transformation Through the Operationalization of Living HTA
Grammati Sarri, PhD, Cytel Inc, London, UK; Seye Abogunrin, MD, MPH, Roche, Basel, Switzerland; Abeer Al Rabayah, MSc, King Hussein Cancer Center, Amman, Jordan; Ajibade Ashaye, MD, MBA, Takeda Pharmaceutical Company Ltd., Cambridge, Massachusetts, USA; Hector E. Castro, MD, DrPH, Access Partnership, Washington DC, USA; Dalia Dawoud, PhD, Cytel Inc, London, UK; Deidre DeJean, PhD, Canada’s Drug Agency, Ottawa, Ontario, Canada; Ramiro Gilardino, Buenos Aires University School of Medicine, Buenos Aires, Argentina; Kun-Tung Hu, MSc, Center for Drug Evaluation, Taipei, Taiwan; Suchitra Iyer, PhD, The Agency for Healthcare Research and Quality, Rockville, USA; Debjani Mueller, DEng, University of Pretoria, Linkenheim-Hochstetten, Germany; Melanie Whittington, PhD, Leerink Center for Pharmacoeconomics, MEDACorp, Boston, MA, USA on behalf of the ISPOR Living Health Technology Assessment Working Group of the ISPOR Health Technology Assessment Council

Health technology assessment (HTA) remains a cornerstone of evidence-based healthcare decision making in many parts of the world—despite differences in political contexts, healthcare financing, and the underlying rationale (local, regional-, or national-level decision making).1 Globally, HTA bodies are confronted with a common set of challenges in carrying out their duties as they assess the value of new health technologies.
We categorize these operational challenges into 3 interrelated domains, each reflecting intrinsic pressures due to evolving technology, rising healthcare costs, and heightened patient and community demands for access. These challenges are linked to the ISPOR Top 10 HEOR Trends for 2026-2027:2
- The need to reliably measure the impact of innovative technologies supported by novel or complex study designs (eg, pragmatic or decentralized trials), insufficient data at the time of assessment, and high levels of uncertainty about their value
(Trends #2, #5, and #9);
- Growing funding and affordability constraints and shocks to health systems from events like the COVID-19 pandemic (Trend #4); and
- Increased demand for accelerated drug approvals and early access to innovative medicinal products (Trend #8).
Collectively, these challenges create a compelling need for a fundamental transformation of HTA that strikes a balance between rapid access and ensuring drug safety and effectiveness. This urgency is further heightened when considering interventions for patient populations with significant unmet needs, such as pediatric populations and those affected by rare and ultra-rare diseases.
The traditional HTA process is long, capturing different interdependent steps, and by the time assessment is conducted, the supporting evidence on which the assessment was originally based may be considered outdated.
Living HTA as a Transformative Approach
The “living” concept in HTA has been developed in response to this need to balance rapid access with drug safety and effectiveness. Living HTA aims to be dynamic, reactive, and agile to facilitate decision making with the most current and relevant evidence in support of changes in policy and practice,3 as it offers a structured way to address evidence uncertainty in a timely manner (Figure).
Figure. Moving from a Static HTA to a Dynamic Living HTA Process
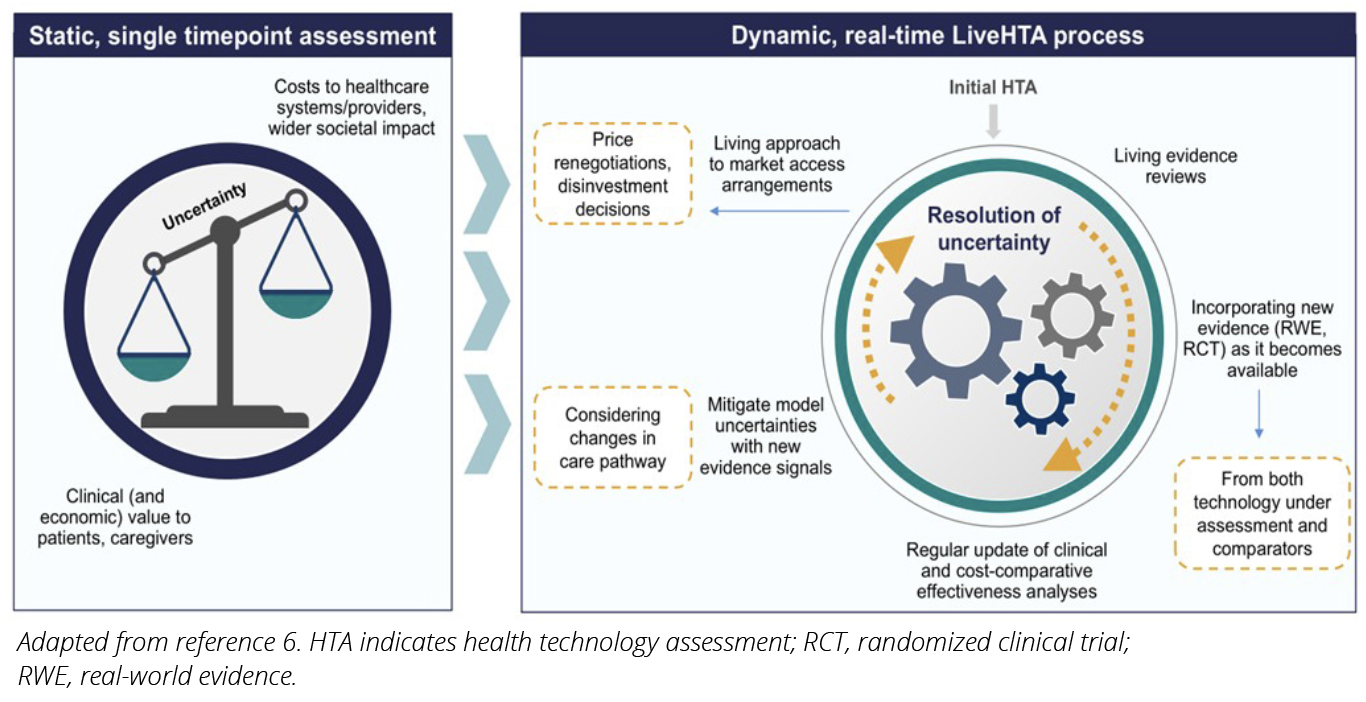
Traditional HTA is often reactive, occurring at a single point in time following initial regulatory approval or in response to regulatory changes (eg, expansion of approved indications). The fundamental activity (with certain exceptions, such as HTAs in low-to-middle income countries [LMICs]) involves an evidence assessment of the emerging technology and its impact on improving patient and/or societal outcomes. The traditional HTA process is long, capturing different interdependent steps, and by the time assessment is conducted, the supporting evidence on which the assessment was originally based may be considered outdated. A 2022 review, for example, identified key structural and methodological issues with the models used by the National Institute for Health and Care Excellence (NICE) for technology appraisals in England and cited these shortcomings as reinforcing the need for regular and consistent technology reassessment.4
Recently, in response to the rapidly evolving evidence base during the COVID-19 pandemic, the concept of decision making with up-to-date information began to be discussed and prioritized.1 This idea of a living HTA gained momentum through the widespread development of living guidelines during the pandemic to provide up-to-date recommendations.5
The proposed definition by the ISPOR Living HTA Working Group6 is: “Living HTA is a systematic, ongoing HTA process that captures the evolving nature of the evidence base, the technology itself, or the dynamic context in which health technologies are adopted leading to an update of the linked recommendation, when needed.”
Some recent examples have demonstrated that a living HTA process can provide faster access to effective health technologies. The Australian Stroke Foundation was able to incorporate new evidence into guidelines in 458 days using the living approach, substantially faster than the 1400 days required with conventional guideline development methods.7 A real-world HTA example in Sweden demonstrated how additional follow-up data can change the direction of decision making, specifically with regard to a novel treatment for Parkinson’s disease. The treatment was originally reimbursed under coverage with an evidence development plan and, following HTA reassessment, it was withdrawn. However, continued evaluation and refinement of the economic model with emerging data demonstrated a clinical benefit, leading to the reinstatement of the technology in clinical practice.8
Living HTA vs Life-Cycle HTA
It is useful to differentiate living HTA from other evolving HTA paradigms, notably life-cycle HTA.9 Life-cycle HTA is a broad concept that uses a planned sequence of HTA activities to inform decisions as the technology, its evidence base, or its clinical context changes over time.10 The core feature of life-cycle HTA is that it prospectively connects and coordinates several distinct HTA activities (such as horizon scanning, scientific advice, and reassessment) that occur at different points in a technology’s lifespan, moving beyond the traditional snapshot assessment at market entry.
While closely related, the living HTA approach proposed in this article can be understood as a specific, highly dynamic implementation of a life-cycle principle. Its defining characteristic is not simply reassessment at preplanned intervals, but an agile and continuous surveillance for triggers that could lead to the initiation of an update as impactful information emerges, making it uniquely suited to the current environment of rapid innovation and real-time data generation. Although fully realized examples of “living HTAs” are still in development, initiatives such as NICE’s living guidelines or CDA-AMC (Canada’s Drug Agency) oncology reassessments already incorporate key life-cycle features, such as modeling sequencing, reassessment triggers, and managed access agreements.11 These examples illustrate how living HTA can serve as a practical mechanism within the broader life-cycle HTA paradigm.
The living HTA approach offers a distinct advantage for health systems with limited resources, such as those in many LMICs. These settings utilize adaptive HTA methods—for instance, adapting an economic model from another country to create an initial, resource-efficient assessment adapted in the local context. The living HTA process enables LMICs to make these otherwise static adaptive assessments dynamic by incorporating emerging evidence for decision making.
Living HTA is a systematic, ongoing HTA process that captures the evolving nature of the evidence base, the technology itself, or the dynamic context in which health technologies are adopted, leading to an update of the linked recommendation when needed.
Operationalization Strategies
Effective operationalization, however, depends on a well-defined framework that supports both methodological structure and procedural flexibility. The process of maintaining a living HTA is not arbitrary but is guided by predefined triggers that would initiate an update. Such triggers could include the publication of pivotal new clinical trial data, the accumulation of a prespecified volume of real-world evidence that challenges original assumptions, significant changes in the standard of care for a disease area, or signals indicating that a technology’s safety or effectiveness profile has shifted in routine practice. A living HTA approach is driven by continuous learning, information sharing, and rapid decision updates, which can facilitate an environment of collaborative innovation between manufacturers and decision makers.
The integration of AI tools in the operationalization of this process will be a key implementation strategy.12 For instance, AI-powered algorithms could be deployed for continuous, automated scanning of new publications and results of real-world data studies, flagging significant new evidence that may trigger an updated assessment of the evidence. Additionally, natural language models could automate the extraction and synthesis of data from clinical trial reports, electronic health records, and peer-reviewed publications, drastically reducing the manual effort required for evidence updates. Furthermore, machine learning could help predict long-term outcomes from surrogate endpoints presented at launch, with models being continuously refined as new real-world data become available. This level of automation will be critical to making a living HTA process feasible and sustainable, provided the tools are demonstrated to be methodologically sound and sufficiently transparent to support decision making.
The transition to a living HTA, underpinned by the systematic use of emerging evidence and potentially enhanced by the transformative power of automation and AI, is the logical and necessary path forward.
A living approach may also be beneficial in assessing the HTA methodology itself, potentially identifying ways to make the process more effective and efficient, such as allowing assessments to be expanded to treatment pathways within a healthcare system rather than individual technologies, considering elements such as appropriateness and affordability. Specifically, a living HTA framework may provide a structured mechanism for disinvestment or price renegotiation. By actively monitoring the real-world performance of a technology against its initial value proposition and against new market entrants, HTA bodies can identify technologies that are no longer cost-effective at their current price.7 This same process allows for the timely reassessment of older, established technologies that may have become less efficient compared to new standards of care, freeing up resources for innovation. This continuous reevaluation ensures that healthcare funds are dynamically allocated to technologies providing the best value, directly tackling the system inertia that leads to financial strain.
A roadmap toward implementing a living HTA approach in real life will require a stepwise, structured developmental strategy:
- Identify
core building blocks and actionable pathways for its adoption in practice
- Identify
and simulate the value of specific triggers for a living HTA approach to be
activated
- Prioritize criteria for its implementation in specific disease
areas/technologies
- Define
the long-term governance of a living HTA, including potential timelines and
criteria for when an assessment “stops living” or transitions to a static
recommendation
- Identify
unique considerations in its implementation based on HTA processes, countries,
or settings, and level of HTA development
- Collaborate with working groups from societies with common interests (eg, ISPOR and Health Technology Assessment International [HTAi]) to diversify the perspectives and knowledge base around this topic and, ultimately, strengthen the implementation strategy of this new HTA approach through cross-institutional validation and broader dissemination.
The paradigm of HTA is at a critical juncture. The pressures of rapid technological innovation, constrained budgets, and the demand for faster access to care have exposed the inherent limitations of a static, point-in-time assessment model. The transition to a living HTA, underpinned by the systematic use of emerging evidence and potentially enhanced by the transformative power of automation and AI, is the logical and necessary path forward.
While the operationalization of such an approach presents challenges requiring clear definitions of triggers, prioritization criteria, and building networks through international collaborations, it promises to make HTA more agile, responsive, and sustainable. By embracing this evolution, the global HTA community can ensure HTA remains a cornerstone of evidence-based decision making, positioned to keep pace with innovation and deliver optimal value to patients and healthcare systems alike.
References
- Oortwijn W, Husereau D, Abelson J, et al. Designing and Implementing Deliberative Processes for Health Technology Assessment: A Good Practices Report of a Joint HTAi/ISPOR Task Force. Int J Technol Assess Health Care. 2022;38(1):e37. doi: 10.1017/S0266462322000198.
- ISPOR 2026-2027 Top 10 HEOR Trends Report. ISPOR—The Professional Society for Health Economics and Outcomes Research. www.ispor.org/Top10Trends. Accessed January 20, 2026.
- Thokala P, Srivastava T, Smith R, et al. Living health technology assessment: Issues, challenges and opportunities. Pharmacoeconomics. 2023;41(3):227-237.
- Daly MJ, Elvidge J, Chantler T, Dawoud D. A review of economic models submitted to NICE’s Technology Appraisal Programme, for treatments of T1DM & T2DM. Front Pharmacol. 2022;13:887298. doi: 10.3389/fphar.2022.887298.
- World Health Organization. COVID-19 Clinical Management: Living Guideline. https://www.who.int/publications/i/item/WHO-2019-nCoV-clinical-2023.2. Published August 18, 2023. Accessed January 20, 2026.
- Living Health Technology Assessment (HTA): HTA Council Working Group. ISPOR. https://www.ispor.org/member-groups/councils-roundtables/health-technology-assessment-council/living-health-technology-assessment. Accessed January 25, 2026.
- Sarri G, Forsythe A, Elvidge J, Dawoud D. Living health technology assessments: How close to living reality? BMJ Evid Based Med. 2023;28(6):369-371. doi: 10.1136/bmjebm-2022-112152.
- Willis M, Persson U, Zoellner Y, Gradl B. Reducing uncertainty in value-based pricing using evidence development agreements: The case of continuous intraduodenal infusion of levodopa/carbidopa (Duodopa®) in Sweden. Appl Health Econ Health Policy. 2010;8(6):377-386. doi: 10.2165/11531160.
- Pichler FB, Boysen M, Mittmann N, et al. Lifecycle HTA: Promising applications and a framework for implementation. An HTAi Global Policy Forum Task Force Report. Int J Technol Assess Health Care. 2024;40(1):e50. doi: 10.1017/S0266462324000187.
- Kirwin E, Round J, Bond K, McCabe C. A conceptual framework for life-cycle health technology assessment. Value Health. 2022;25(7):1116-1123. doi: 10.1016/j.jval.2021.11.1373.
- Cupples G, Krebs E, Weymann D, Ho C, Regier DA. Adopting life-cycle HTA: A tumor-agnostic precision oncology index economic evaluation from publicly available reimbursement reviews. Int J Technol Assess Health Care. 2025;41(1):e41. doi: 10.1017/S0266462325100111.
- Fleurence RL, Bian J, Wang X, et al. Generative artificial intelligence for health technology assessment: opportunities, challenges, and policy considerations: An ISPOR Working Group Report. Value Health. 2025 Feb;28(2):175-183. doi: 10.1016/j.jval.2024.10.3846.

