Unlocking the Voice of Patients: Are Engagement Initiatives Transforming Canadian HTA Reviews?
Nicole Fusco, ScD; Brittany Galloway, PharmD; Malia Gill, MS; Sarah Cadarette, MPH; Erika Wissinger, PhD; Maria Koufopoulou, MS, Cencora, Conshohocken, PA, USA

Introduction: The Need for Patient Centricity in Clinical Trials
Traditionally, the evidence supporting drug treatment benefit primarily relied on clinical endpoints, but since the early 1990s, understanding of the importance of incorporating the “patient voice” into the assessment of treatment efficacy and effectiveness has grown. Ultimately, the patient holds the best perspective of their experiences with a particular health state and treatment. While clinical outcome measures provide what are considered “hard,” unequivocal outcomes (eg, mortality, lab values, tumor shrinkage), they may not capture factors that impact the patient’s quality of life and matter most to the patient. In some cases, the patient experience may differ from treatment effects described by clinical outcomes; to the patient, for example, negative side effects of treatment may outweigh apparent clinical benefits. Thus, the patient experience is critical to understanding and comparing the benefits and risks of treatment.
When treatments have similar primary clinical outcome results (eg, similar survival rates), data from patient-reported outcomes (PROs) can provide information about the impact of treatment on symptoms of the underlying disease, possible side effects, and how those experiences impact a patient’s daily activities.1 Results from PRO data may align with other clinical outcomes (eg, improvement in survival corresponding with improved symptoms), or they may provide contrasting results (eg, improvement in survival but decreased health-related quality of life [HRQoL]). Moreover, some progressive diseases, such as rheumatoid arthritis, lack objective patient-relevant clinical endpoints. Developing and utilizing standardized PRO measures allows healthcare providers and researchers to assess patient symptoms, functioning, and HRQoL in ways that can be compared across groups of patients (eg, in clinical trials) or over time (eg, to assess patient response to treatment).2
Over the past 25 years, regulatory bodies such as the US Food and Drug Administration and the European Medicines Agency have become increasingly focused on taking the patient perspective into account when evaluating drug benefits. Similarly, health technology assessment (HTA) agencies have developed their own patient engagement strategies to better understand the value of treatments. Although HTA reviews are conducted separately from regulatory reviews, both are typically based on the same registrational trials, amplifying the impact of decisions made during trial design.
We sought to assess how Canada’s Drug Agency (CDA-AMC) has included the patient perspective in its HTA reviews and how increasing emphasis on the patient experience has impacted the data available to inform this evaluation.
How Does CDA-AMC Involve Patients in the Reimbursement Process?
Under CDA-AMC, patient involvement in the reimbursement process has been relatively straightforward (Figure 1). Prior to conducting a review, CDA-AMC solicits input from patient groups, summarizes that input, and validates it with the patient groups. Following the review, patient feedback is requested on the recommendations. Patient input generally covers their experience with the disease and currently available treatments, unmet needs, and outcomes that are important to patients. In the methodology guidance for HTA reviews, CDA-AMC states that the outcomes of interest should include “clinically meaningful endpoints such as mortality, morbidity, and patient-reported experiences, symptoms, health behaviors, function, and HRQoL.”3 Additionally, in January 2026, CDA-AMC announced its updated patient group input process, including a revised Patient Group Input Template designed to align patient input with the 5 domains of value: clinical value, unmet clinical need, distinct social and ethical considerations, economic considerations, and impacts on health systems.4
Figure 1. CDA-AMC patient involvement
Do Reimbursement Reviews Include the Data That Really Matter to Patients?
In our evaluation of HTAs submitted to CDA-AMC, we focused on drug treatments for 3 conditions: non-small cell lung cancer, asthma, and rheumatoid arthritis. The 3 conditions were chosen because they exemplify the continuum of ways in which the subjective patient experience is incorporated into clinical outcomes.
- The main clinical outcomes used in assessing therapies for rheumatoid arthritis rely heavily on patients’ evaluations of their pain, ability to carry out daily activities, and global disease assessment.
- Clinical outcomes for asthma are more objective measures of lung function, but patient-reported symptoms are also commonly included.
- Non-small cell lung cancer clinical outcomes focus on objective measures of survival and response.
Most trials included in the CDA-AMC reimbursement reviews on treatments for non-small cell lung cancer (n=55 trials), asthma (n=37 trials), and rheumatoid arthritis (n=24 trials) reported at least some PRO data, although the types of PROs varied by condition (Figure 2).
Figure 2. Types of outcomes reported in trials in CDA-AMC reimbursement reviews
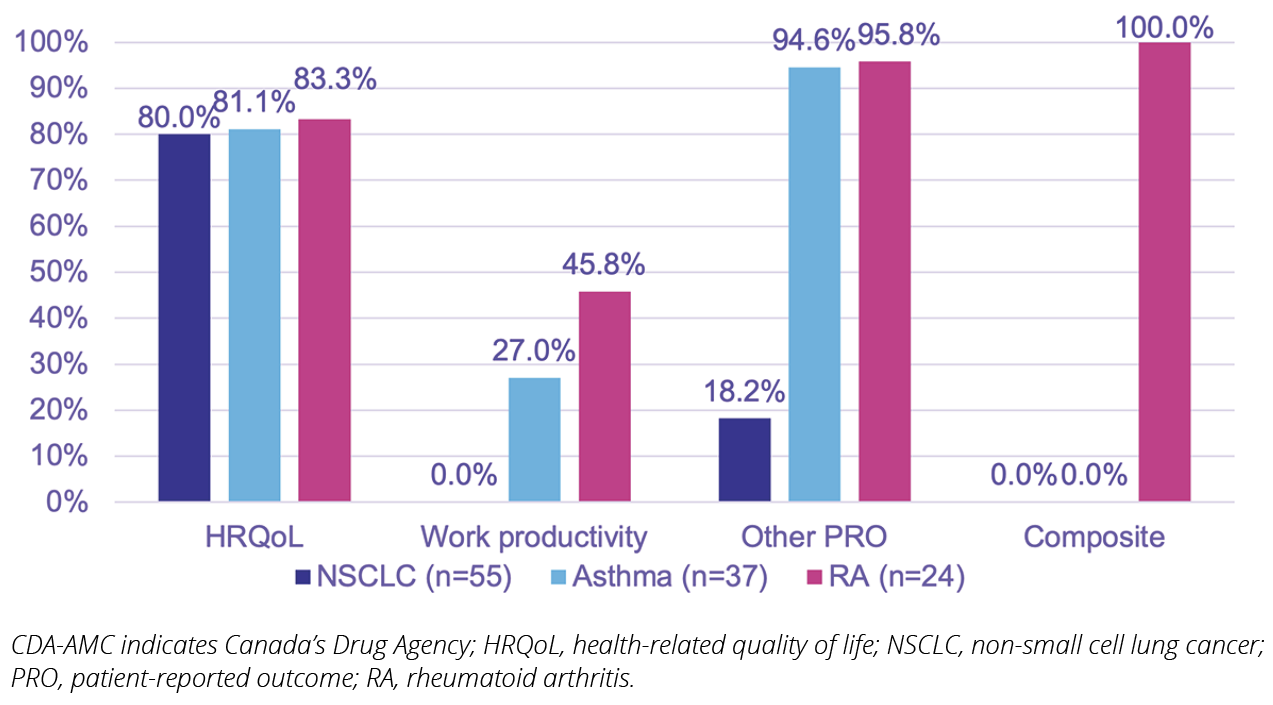
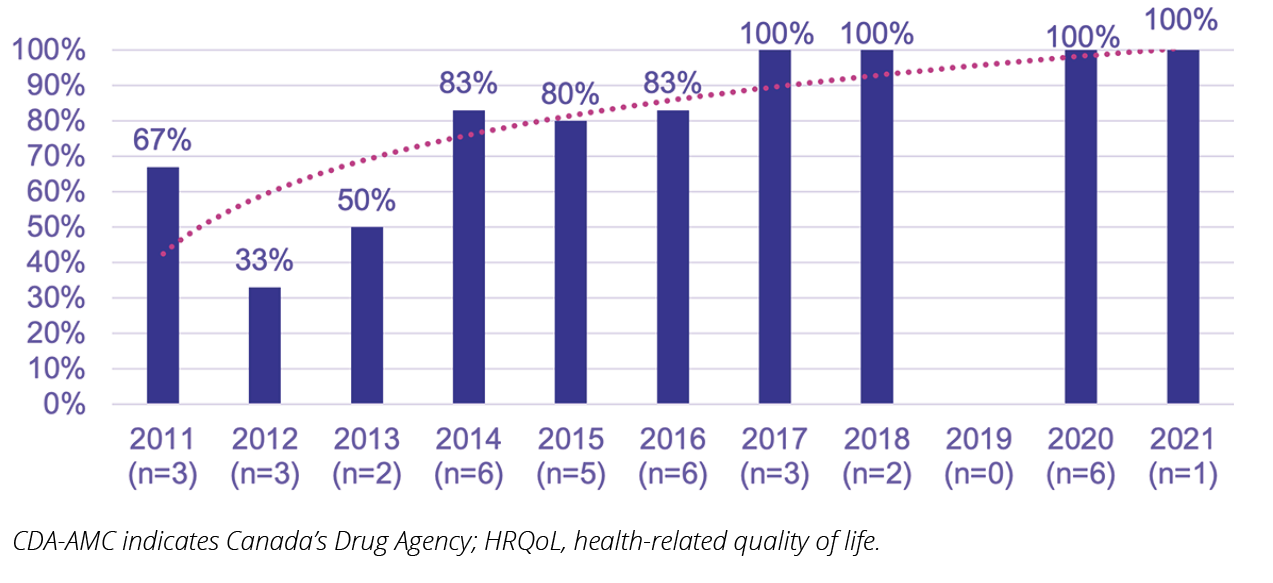
The PROs captured in these trials have been categorized here as HRQoL, work productivity, composite outcomes (ie, outcomes that include some patient-reported information [such as pain] combined with clinician assessment, global disease assessment, and/or purely objective measures such as lab values), or other PROs (eg, specific disease symptoms, global assessments of disease, satisfaction with treatment). Across the indications, HRQoL was the most common type of PRO assessed; all rheumatoid arthritis trials also included at least one composite outcome (most commonly the American College of Rheumatology response criteria). Nearly all asthma and rheumatoid arthritis trials included at least one other PRO, typically assessing specific symptoms such as pain, fatigue, wheezing, or coughing. Additionally, patient-reported work productivity has been incorporated into some asthma and rheumatoid arthritis trials. Although the number of trials each year was small, the inclusion of HRQoL in asthma trials has increased over time (Figure 3).
Figure 3. Asthma trials in CDA-AMC reimbursement reviews that included HRQoL measures by year
What Should We Target Next?
The results of our exercise indicate that HRQoL was assessed in the majority of non-small cell lung cancer, asthma, and rheumatoid arthritis trials included in CDA-AMC reviews. The specific PRO measures used across studies varied; some used generic HRQoL scales (eg, EQ-5D), while others used disease-specific scales.
In the 30 asthma trials that assessed HRQoL, most used the Asthma Quality of Life Questionnaire. However, there was variation in the version of the questionnaire that was used. When looking at the tools used to assess symptoms in asthma trials, the landscape is even more heterogeneous.
Although there are methods for quantitatively comparing different tools used to assess the same construct, these methods often reduce interpretability of the data.5 In addition, some of the methods (eg, using a threshold to convert continuous measures to dichotomous outcomes) can only be applied with individual patient data from the original trials, which are often inaccessible to many systematic reviewers and other stakeholders.
Without specific guidance from regulatory and reimbursement agencies, trial sponsors may have difficulty determining the best, most relevant outcomes to collect.
There have been efforts to develop core outcome sets (ie, a standardized set of outcomes that should be collected in all new trials), for many disease areas, including asthma.6 However, adoption of core outcome sets is still somewhat limited and depends heavily on the disease area.7 For some disease areas, there are dozens of published core outcome sets, resulting in the same problem on a different scale.
How Do We Get There?
Much of the improvement in the collection of HRQoL and other PRO data can likely be attributed to guidelines from regulatory and reimbursement agencies, as well as encouragement from professional societies such as the American Society of Clinical Oncology.8-10 Although guidance from regulatory and reimbursement agencies may help to standardize outcome collection across trials, it would be burdensome to generate specific outcome guidance for every disease area. Conversely, creating broad guidance might limit relevance of the outcomes to the specific condition of interest.
Some regulatory agencies have established programs to promote up-front communication between sponsors and regulatory agencies, allowing them to provide guidance on the collection of PROs early on in trial design. As regulatory and HTA agencies increase patient engagement initiatives, patient organizations have the opportunity to encourage the use of core outcome sets developed with patient involvement. Patient organizations can also influence the uptake of core outcome sets through involvement with clinical guideline development or directly with trial sponsors.7
Increased interest in the patient perspective reinforces the need to treat patient-reported outcomes as a core component of evidence generation.
Encouragement from professional societies may influence outcome collection, but without the same kind of authority as regulatory and reimbursement agencies, these types of initiatives may fall short. Without specific guidance from these agencies, trial sponsors may have difficulty determining the best, most relevant outcomes to collect. Conducting literature reviews early on in trial design may be an efficient method to identify relevant core outcome sets, PRO measures regularly used in similar trials, and information about the validity, reliability, clinical significance, and ability to detect meaningful change using those PRO measures. This information can also be utilized in regulatory and reimbursement submissions to facilitate the agency’s review of the instrument.
What Are the Practical Implications for HTA Submissions?
Although these results are specific to CDA-AMC reviews, other HTA agencies have their own patient engagement initiatives, indicating that PROs and the patient experience are of increasing interest across agencies. In addition, submissions to various HTA agencies typically utilize data from the same registrational trials, meaning that increased levels of patient engagement across HTA agencies are working synergistically to yield additional PRO data to inform reimbursement decisions beyond the Canadian context.
Pragmatically, this increased interest in the patient perspective reinforces the need to treat PROs as a core component of evidence generation. Because HTA dossiers in multiple jurisdictions often draw from the same registrational program, early, deliberate selection of PRO instruments (including consideration of existing core outcome sets where available) can improve cross-trial comparability. In the submission itself, clearly articulating the rationale for the selected concepts and instruments, linking them to the outcomes that patients say are most important, and prespecifying how PRO data will be analyzed and contextualized alongside clinical endpoints can support more transparent benefit–risk and value discussions during HTA review, even when clinical outcomes may appear similar across treatments.
References
- Au HJ, Ringash J, Brundage M, Palmer M, Richardson H, Meyer RM. Added value of health-related quality of life measurement in cancer clinical trials: the experience of the NCIC CTG. Expert Rev Pharmacoecon Outcomes Res. 2010;10(2):119-128. doi:10.1586/erp.10.15
- Johnston BC, Patrick DL, Busse JW, Schünemann HJ, Agarwal A, Guyatt GH. Patient-reported outcomes in meta-analyses – Part 1: assessing risk of bias and combining outcomes. Health Qual Life Outcomes. 2013;11(1):109. doi:10.1186/1477-7525-11-109
- Canada’s Drug Agency (CDA-AMC). Methods Guide for Health Technology Assessment. https://www.cda-amc.ca/methods-guide. Published March 27, 2025. Accessed April 7, 2026.
- Canada’s Drug Agency (CDA-AMC). New Patient Group Input Template: Improving How Patient Perspectives Inform Drug Reimbursement Reviews. https://www.cda-amc.ca/news/new-patient-group-input-template-improving-how-patient-perspectives-inform-drug-reimbursement. Published January 29, 2026. Accessed April 10, 2026.
- Johnston BC, Patrick DL, Thorlund K, et al. Patient-reported outcomes in meta-analyses -Part 2: methods for improving interpretability for decision-makers. Health Qual Life Outcomes. 2013;11(1):211. doi:10.1186/1477-7525-11-211
- Khaleva E, Rattu A, Brightling C, et al. Development of Core Outcome Measures sets for paediatric and adult Severe Asthma (COMSA). Eu Respir. 2023;61(4):2200606. doi:10.1183/13993003.00606-2022
- Williamson PR, Barrington H, Blazeby JM, et al. Review finds core outcome set uptake in new studies and systematic reviews needs improvement. J Clin Epidemiol. 2022;150:154-164. doi:10.1016/j.jclinepi.2022.06.016
- Mercieca-Bebber R, King MT, Calvert MJ, Stockler MR, Friedlander M. The importance of patient-reported outcomes in clinical trials and strategies for future optimization. Patient Relat Outcome Meas. 2018;9:353-367. doi:10.2147/PROM.S156279
- Guidance for Industry Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims. US Food and Drug Administration. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/patient-reported-outcome-measures-use-medical-product-development-support-labeling-claims. Published December 2009. Accessed April 7, 2026.
- Appendix 2 to the guideline on the evaluation of anticancer medicinal products in man. The use of patient-reported outcome (PRO) measures in oncology studies. European Medicines Agency. https://www.ema.europa.eu/en/appendix-2-guideline-evaluation-anticancer-medicinal-products-man-use-patient-reported-outcome-pro-measures-oncology-studies-scientific-guideline. Published April 22, 2016. Accessed April 7, 2026.
The information provided herein does not constitute legal advice and the authors encourage readers to review all available information related to the topics discussed, including any references provided, and to rely on their own experience and expertise in making decisions related thereto.

