Predicting PICOs Under EU Joint Clinical Assessment: Is One Method Enough?
Pamela Vo, MS, PharmD, EVERSANA, Basel, Switzerland; Niklas Hedberg, MSc, TLV, Stockholm, Sweden; Michael Happich, DrPH, PhD, Eli Lilly, Bad Homburg, Germany; Zsombor Zrubka, MBA, PhD, MD, Obuda University, Budapest, Hungary; Carl Asche, MBA, MSc, PhD, University of Utah, Salt Lake City, UT, USA; Shivani Shah, MPharm, EVERSANA Mumbai, India

The new European Union (EU) Health Technology Assessment (HTA) Regulation (EU 2021/2282) marks one of the most significant changes in European market access in recent decades. The new process, launched in 2025, requires oncology and advanced therapy medicinal products (ATMPs) to undergo Joint Clinical Assessments (JCAs) at the EU level, followed by orphan medicines in 2028 and all new medicinal products by 2030. For the first time, a single clinical assessment will help inform pricing and reimbursement decisions across all EU Member States.
JCA is conducted in parallel with the existing assessment process used by the European Medicines Agency (EMA) to recommend marketing authorization by the European Commission. The JCA report created at the end of each assessment, which summarizes the evidence on the relative clinical effectiveness and safety of a health technology, is intended to help national Member State authorities make more timely, efficient, and informed decisions about allocation of healthcare resources.
At the heart of the JCA process lies PICO (Population, Intervention, Comparator, and Outcomes), factors that comprise the framework within which health technology developer (HTD) submissions are assessed. Every JCA goes through a scoping process to specify PICOs that reflect the requirements of the individual Member States, but these are rarely consistent across countries. Subpopulations differ, comparators evolve and vary across dynamic treatment landscapes, and clinical outcomes may be interpreted differently by national HTA agencies. Consequently, HTDs’ ability to anticipate PICOs before the JCA scoping phase could profoundly shape the evidence strategies, trial design alignment, and dossier complexity.
During ISPOR Europe 2025, panelists from academia, industry, and HTA bodies explored different strategies for PICO prediction and considered a critical question: Is there one best method, or is a blended strategy the only practical path forward?
This article will present the highlights of that discussion, including an overview of the strengths and limitations of the various strategies. It will also outline key practice‑informed early lessons from the first year of JCA implementation that can help HTDs navigate the JCA process.
Misalignment of evidence package submission with final PICOs required for Joint Clinical Assessment could result in costly rework, weak evidence supporting, and/or delays to access.
The Regulatory Shift: From National to EU-Level Assessment
Historically, HTA requirements have varied significantly across individual EU Member States. The EU HTA Regulation introduces a structured, permanent joint framework designed to harmonize clinical assessments across the Member States (while reducing duplication), enhance transparency, and accelerate patient access to innovative therapies in Europe.
Why it matters:
- It establishes a single EU-level submission for clinical assessment.
- It is closely aligned with EMA timelines, with scoping starting early, often before full regulatory clarity (eg, changes to the Summary of Product Characteristics [SmPC] may occur during the EMA process, as mentioned in the next section).
- It has the potential to streamline market access and minimize delays in reimbursement decisions, and
- It raises the stakes: Misalignment of evidence package submission with final PICOs could result in costly rework, weak evidence supporting, and/or delays to access.
The complicated process of harmonizing clinical assessments across all member states, in turn, increases the complexities of establishing consolidated PICOs to facilitate those assessments. It also creates challenges for health technology developers trying to anticipate those PICOs and plan their evidence generation accordingly.
- PICO variability across Member States: Heterogeneity in how Member States define patient groups, comparators, and outcomes leads to significant variation in PICOs. This variability makes the consolidated PICOs difficult to predict and creates substantial uncertainty for HTDs when planning evidence generation, particularly because national needs and requirements often differ across Member States.
- Potential change of SmPC: Recent academic research indicates that up to 33% of ATMPs undergo indication changes during the EMA regulatory process, which could affect the JCA scoping process. These adjustments require rapid adaptation to PICO definitions, potentially affecting trial alignment.
- Evidence generation challenges for specific health technologies:
Products such as oncology therapies, orphan drugs, and gene therapies face persistent evidence challenges—from rapidly evolving standards of care (eg, oncology) to small trial populations, ethical barriers to randomized controlled trials, and greater reliance on heterogeneous real‑world data (eg, orphan drugs/ATMPs). These hurdles create significant uncertainty in meeting JCA evidence expectations.
- Insufficient resource allocation: The harmonized assessment process will require HTDs to allocate resources efficiently to meet the demands of both regulatory and HTA evaluations. In particular, the JCA process demands extensive and detailed data and, therefore, requires significant resources and coordination due to the tight deadlines for dossier submissions.
Sources of evidence for early PICO formulation include:
- Draft SmPC and clinical overview (Marketing Authorization Application file)
- EU and non-EU clinical guidelines
- HTD-driven PICO surveys among Member States
- Reimbursement/HTA documents for products approved in the indication for which PICOs are defined
- JCA guidance documents on scoping and outcomes
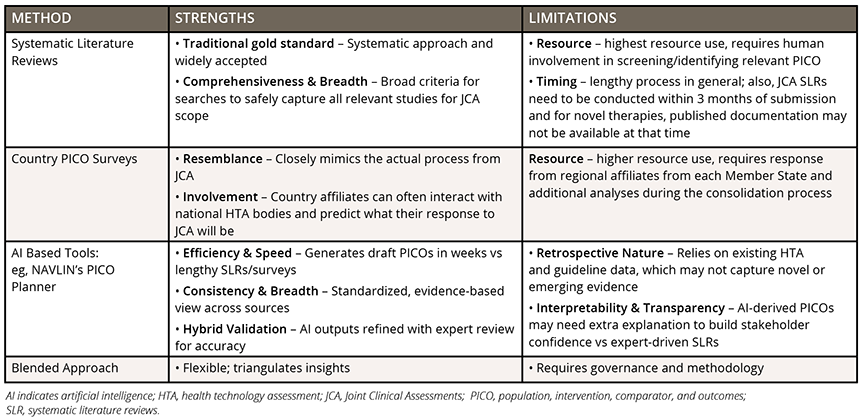
Methods for Predicting PICOs: Strengths and Limitations
As the EU HTA framework moves from concept to implementation, HTDs are increasingly exploring methods to anticipate likely PICOs before the formal JCA scoping phase. Early prediction can help align clinical development, evidence generation, and value demonstration strategies with anticipated assessment requirements. However, no single methodology currently offers a complete solution.
Traditional approaches such as systematic literature reviews and country-level consultations remain widely used but are often resource-intensive and time-consuming. At the same time, emerging data-driven and artificial intelligence-supported tools (eg, NAVLIN AI PICO Planner) are being explored to accelerate and standardize early PICO identification. In practice, many organizations are considering whether a single method is sufficient or whether a blended approach may provide a more robust strategy.
Table 1 summarizes commonly used approaches for early PICO prediction and highlights their key strengths and practical limitations.
Implications for Industry Stakeholders
- Incentive to predict PICOs early and incorporate into clinical trial program, but risk if misaligned
- Early strategic evidence planning to support both regulatory and HTA/payer needs (eg, 12-18 months pre-JCA submission)
- Evidence generation may need to become modular and adaptive
- Resource allocation decisions may shift from “1 trial = 1 PICO strategy” to “1 trial = multiple PICO contingencies”
- The question of which PICO prediction method is best for JCA is less a question of “either/or” but “when and how much”, as all approaches require resources, especially if done repeatedly, to stay up to date
- The EU HTA Regulation requires HTDs to be overinclusive in their PICO scenario planning to prepare for contingencies
- An earlier (joint) agreement on the PICO scope would help mitigate this burden
Implications for HTA Stakeholders in Member States
The JCA is intended to inform pricing and reimbursement decisions in each Member State, and each nation has representatives on the HTA Coordination Group that works with stakeholder groups to jointly define the scope of JCA reports, including PICOs. The HTA agencies from each Member State will want to ensure the PICOs in each JCA meet their nation’s specific clinical and economic needs, which will involve some of the same prediction methods used by health technology developers.
Implications for HTA agencies include:
- Requires a balance between flexibility and standardization
- Need for efficient handling of subpopulations and comparators
- Expressing clear expectations (eg, through joint scientific consultations between health technology developers and JCA advisors) will be essential
- Opportunity to evaluate emerging prediction methodologies
- Role in building validation frameworks, especially for AI solutions
- Can serve as neutral domain experts during discussions of comparator relevance
- The challenge will spur innovation in the method of generating and synthesizing evidence
- The pressure of JCA will drive a tighter integration of HTA and value planning in the development of health technologies.
Many companies are beginning to treat PICO prediction as an early phase activity rather than a postregulatory task. However, consolidating PICOs is not straightforward.
Panelists’ Perspective
The transition to EU-level HTA moves beyond coordination and redefines strategic evidence planning. The scoping phase, historically seen as procedural, is now a pivotal decision point that influences trial outcomes, evidence strategy, and resource allocation. Many companies are beginning to treat PICO prediction as an early phase activity rather than a postregulatory task.
However, consolidating PICOs is not straightforward. Member States differ in treatment practices and clinical guidelines, leading to divergent views. A flexible and evidence-based strategy for PICO prediction appears preferable, but the exact methodology remains unclear.
Key takeaways from the ISPOR Europe panel included:
- Early PICO prediction matters. All panelists agreed that early prediction of PICO elements is vital. This enables HTDs to initiate and plan evidence generation sooner, reducing the risk of gaps and delays later in the process. No single methodology for PICO prediction currently offers a complete solution; rather, hybrid approaches may be needed.
- Continuous learning and adaptation. The JCA process is new for everyone, and early implementation will bring challenges. Applying lessons quickly will be key to building confidence and improving efficiency.
- A shared commitment to success. Stakeholders across the board want the EU HTAR and JCA to succeed—not just as a regulatory milestone, but to deliver faster and broader access to innovative medicines for patients across Europe.
- Collaboration is critical. Strong collaboration among regulators, HTA bodies, and industry will be essential. In particular, HTDs suggest that more proactive engagement between them and the Member State HTA Coordination Group (established by the Regulation) can help ensure evidence generation aligns with evolving requirements.
While companies continue to navigate evolving expectations and operational complexity, the first year has also demonstrated opportunities to embed JCA considerations earlier in development and evidence-generation plans.
The First Year: The Consultant Perspective
From the perspective of the consultancy supporting HTDs, the first year of JCA implementation has served as a learning and adaptation phase for both industry stakeholders and assessors. Working closely with companies across multiple JCAs has underscored the critical importance of early strategic planning, evidence alignment, and clear articulation of clinical value at the EU level.
While companies continue to navigate evolving expectations and operational complexity, the first year has also demonstrated opportunities to embed JCA considerations earlier in development and evidence-generation plans. These early lessons reinforce the need for continued methodological clarification and practical guidance by the HTA Coordination Group to help translate JCAs into efficient and meaningful national HTA and access outcomes.
In particular, the first year of JCA implementation has highlighted several critical success factors for navigating the EU HTA Regulation effectively:
- Tailor PICO prediction approaches by therapeutic area or timing. In practice, the most effective strategy for anticipating PICOs may involve combining multiple approaches and tailoring them to the therapeutic context or stage of development.
- For example, in therapeutic areas such as oncology, where standards of care evolve rapidly and comparator landscapes differ across Member States, faster approaches such as AI-based tools can help identify likely comparators and patient subpopulations early in development. These early insights can then be validated through systematic literature reviews closer to submission, when more published evidence becomes available.
- For rare disease areas, PICO surveys with country affiliates or external validation with key opinion leaders and healthcare professionals may add more value than AI-based tools that rely primarily on retrospective evidence.
- Similarly, when a drug is in the early development phases (phase I or phase II), AI-based tools can support competitive landscape analysis and help identify potential PICOs. As the product progresses into phase III, PICO surveys and systematic literature reviews can further support the development of evidence-based PICOs aligned with anticipated JCA requirements.
- Allocate appropriate budget and resources early. Preparing a robust JCA dossier is highly resource-intensive and evidence heavy, requiring substantial cross-functional input, analytical rigor, and coordination across global and European teams. Under-resourcing JCA preparation risks downstream inefficiencies and limits the ability to respond effectively to assessor questions within compressed timelines.
- Establish clear governance, operating models, and realistic expectations. Successful JCA execution requires early definition of governance structures, decision-making accountability, and roles across global, regional, and local teams. Managing internal expectations, particularly around timelines, flexibility, and the relationship between JCA outputs and national HTA needs, has emerged as critical to maintaining alignment and momentum.
- Plan evidence strategically and early to enable execution. Early and proactive evidence planning, aligned with anticipated JCA scope and comparators, is essential. The first year has underscored the importance of embedding JCA considerations into clinical development and evidence-generation plans well in advance, allowing sufficient time for PICO assessment, data readiness, analyses, and dossier development.
- Leverage external expertise to support strategic positioning. Involving external experts (including clinical experts, health economics and outcomes research (HEOR) experts, and HTA-experienced advisors) can enhance the robustness, credibility, and clarity of JCA submissions. External perspectives can also help challenge assumptions and strengthen value narratives for a multistakeholder assessment environment.
- Balance broad cross-functional collaboration with agility in decision making. While effective JCA preparation requires close collaboration across clinical, regulatory, HEOR, market access, and legal teams, the first year has shown the importance of remaining agile. Clear escalation pathways and decisive leadership are essential to avoid delays and ensure timely alignment on evidence strategy and responses.
Closing Remarks
The HTA Regulation represents a transformative shift toward harmonization and equity in patient access. While the journey will require flexibility and collaboration, the potential benefits (eg, greater consistency, transparency, and speed) make this an exciting time for the HEOR field.

