Industry Stakeholders Reflect on Patient Centricity’s Progress and Prospects for a More Equitable Future
Matthew Reaney, PhD, CPsychol, CSci, IQVIA Patient Centered Solutions, London, UK

Biopharmaceutical medicines development has long been focused on scientific innovation. This is essential and has led to the eradication of many diseases as well as significant improvements in the day-to-day clinical management of chronic diseases. However, medicines only work in people who choose to take them,1 and the desire and drive to answer questions like “What can be done?” in medicines development sometimes led to avoidance of answering questions like “What should be done?”.
As a health psychologist, I think about “what should be done” in 2 ways: What is the burden of disease for patients and healthcare systems, and What are the patient and clinician priorities, preferences, and needs in managing that disease? Inclusion of the patient is essential.
Patient Centricity in Medicines Development
Calls for a more patient-focused approach to medicines development have been growing from governments, insurers, and patient organizations for years, and the biopharmaceutical industry is embracing them through the “patient centricity” movement. Companies are now working more closely with patient advocacy groups, and company mission statements have been reworked to reflect a commitment to improving lives.2
However, in 2020, a survey of more than 1000 patients from the United Kingdom and Ireland showed that the biopharmaceutical industry is one of the least trusted industries.3 One reason was because survey respondents believed the approach and priorities of pharmaceutical companies were driven by profits rather than the needs of patients—that companies were not truly committed to the principles of patient centricity but instead were skirting around the idea by starting a few highly visible initiatives without weaving it into the fabric of the organization.
Further reflection and discussion are, therefore, needed.
Survey data suggest one reason for lack of trust in the biopharmaceuticals industry is the belief that companies are driven by profits rather than the needs of patients.
Collecting Industry Perspectives on Patient Centricity
In early 2025, I asked more than 30 people who work in and around the biopharmaceutical industry for their thoughts on patient-centric intervention development. Specifically, I invited pharmaceutical company executives, small biotechnology manufacturers, patient representatives/patient advocates with lived experiences, patient experience data (PED) researchers, regulatory representatives, and payer and health technology agency (HTA) advisors to reflect on where we are today as an industry, what improvements are still to be made, and whether patient centricity offers a genuine shift for the future of healthcare. They could offer their thoughts through a personal narrative, a review of the research, a poem, a series of drawings, or whatever made sense to them.

The contributions were collated and bound in a book in April 2025.4 Only stylistic editing has been applied to their musings and the views presented are therefore somewhat unfiltered and honest, grounded in the authors’ own experiences, knowledge of and/or participation in research, and hopes for the future. See Figure 1 for a list of contributor organizations.
A summary of findings from across the contributions was presented at the ISPOR Europe Congress 2025, where the topic was “powering value and access through patient-centered collaboration.”5 This summary is also presented below.
Figure 1. Contributor organizations

Findings From the Collection
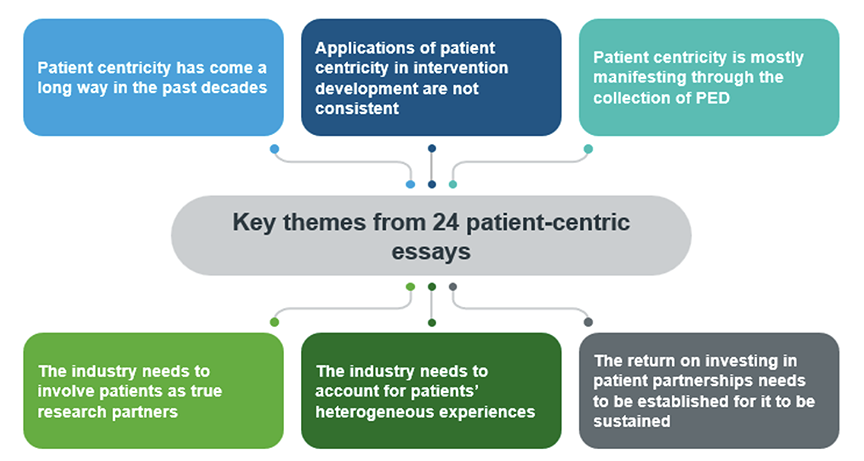
Six clear themes have emerged from the contributions to the collection (see Figure 2).
1. We’ve Come a Long Way.
Not long ago, medicines development rarely involved patients. Products were designed around scientific innovation and researcher convenience, with little regard for patient priorities or experiences. Clinical trials often excluded patient input, and outcomes focused on clinical metrics such as disease progression or biomarkers rather than symptomatic improvement such as reduction in pain or cough, and quality of life.
Thankfully, this has changed. Advances in technology, shifting societal attitudes, and growing patient advocacy have pushed the industry to recognize that successful treatments must be acceptable to both clinicians and patients, in the same way as any consumer product must be acceptable (if not desirable) to the end user or purchaser. Today, it is an expectation—at least in the United States and Europe—and many companies actively seek patient input to ensure new interventions meet real needs. This is particularly true for rare diseases, where heterogeneity in patient experience must be understood to design trials that patients are able to participate in and with measure outcomes that they care about.
2. Patient Centricity Is Still Inconsistently Applied.
While patient-centered medicines development is now expected in the United States and Europe, its application varies widely. Contributors to this collection offered definitions ranging from “listening to patients” to “designing meaningful and accessible interventions.” Despite this shared intent, the practical implementation remains inconsistent. True patient centricity is difficult to achieve, especially within the rigid frameworks of clinical trials and regulatory requirements. Too often, patient involvement is limited to short-term consultations that feel more like box-ticking than genuine collaboration. This superficial engagement risks undermining trust in an industry that claims to prioritize patients.
True patient centricity is difficult to achieve, especially within the rigid frameworks of clinical trials and regulatory requirements.
3. Patient Experience Data Is the Main Avenue.
The most common way patient centricity is expressed today is through the collection of PED in clinical trials. PED includes insights into how patients feel, function, and experience their condition and its treatment. The most widely used form of PED is patient-reported outcomes (PROs), which measure symptoms, quality of life, and treatment tolerability from the patient’s perspective. These data can inform regulatory decisions, support product labeling, and guide clinical care. However, PROs offer only part of the picture. To truly understand patient experiences, researchers must combine qualitative and quantitative methods—and listen deeply to what matters most to individuals.
4. The Biopharmaceutical Industry Needs to Involve Patients as True Research Partners.
Patient centricity encourages companies to place patients at the center of drug development—but patients don’t want to be the sole decision makers. They want to be equal partners alongside clinicians, regulators, and payers. Most patients seek a meaningful voice in shaping interventions, recognizing that their priorities may not always align with clinical or regulatory constraints.
An equitable model where the patient voice is as valued as other stakeholders is emerging as the optimal approach. This partnership should begin early and continue throughout the development process. Technology, including artificial intelligence and online platforms, can help broaden and sustain these collaborations.
Partnerships should start by identifying patient needs and preferences. Patient partners can then provide feedback on potential treatments, help design clinical programs (including which outcomes to measure to demonstrate value), and shape trials to reduce participant burdens and improve relevance. Their involvement can also support recruitment, training, and interpretation of results. Post trial, patient partners can help communicate findings and contribute to regulatory submissions and educational materials, as well as design real-world data collection approaches such as registries or observational studies.
To make this model work, companies must embed patient involvement as a core value, supported by standardized frameworks and cross-functional objectives. Education and training—for both industry professionals and patients —are essential to foster effective collaboration.
One novel example of this in a pharmaceutical company involved a Global Head of Patient Centricity overseeing teams focused on integrating the patient perspective in discovery and early development as well as in a real-world setting, in addition to teams focused on culture and integration, partnership, and behavioral science.6
Most patients seek a meaningful voice in shaping interventions, recognizing that their priorities may not always align with clinical or regulatory constraints.
5. Patient Partnerships Must Account for Heterogeneous Experiences.
Not all patients have the same experiences, and one voice cannot represent all. Contributors to the collection highlighted the lack of diversity in drug development and called for broader inclusion across race, ethnicity, socioeconomic status, geography, and health literacy. Without this, interventions risk being irrelevant to large segments of the population.
Partnering with patient advocacy groups and charities can help reach underrepresented communities and ensure more inclusive research. While regulators like the US Food and Drug Administration have pushed for diversity in trials in the past, recent political shifts have challenged these efforts. These evolving dynamics have increased the need for companies to continue to prioritize diverse perspectives to ensure treatments serve society as a whole.
6. The Return on Investment in Patient Partnerships Needs to Be Established.
While regulators in the United States and Europe encourage patient-centric development, it’s not a global mandate. Contributors agreed it’s the right thing to do—ethically and practically—but acknowledge that without clear return on investment (ROI), companies may hesitate to fully commit.
True partnership requires time, resources, and structural changes. The benefits—such as improved product relevance and trust—are real but not always easy to quantify. Some efforts have been made to quantify the ROI of patient engagement for the biopharmaceutical industry, but they have largely been conceptual.
For example, a study by the Clinical Trials Transformation Initiative, in collaboration with Tufts Center for the Study of Drug Development and Janssen, suggested that a modest $100,000 investment in patient engagement may result in savings of up to $2.1 million in avoidance of one substantial protocol amendment in a phase III oncology trial.7 Other research has suggested that enrollment and retention in clinical trials can be enhanced through PROs.8 Some progress has also been made in quantifying the ROI for patient engagement beyond clinical trials, like the inclusion of patient-reported outcomes in drug labels and commercialization,9,10 but findings are not conclusive or particularly persuasive.
Without clear ROI metrics, patient centricity risks becoming a checkbox exercise. To embed it meaningfully, companies need models that track impact from development through approval, reimbursement, and market uptake.
Figure 2. Key themes across the contributions
One Year Later
It is now nearly 1 year since the publication of the book that provided these insights from across the industry. In that time, I have attended multiple patient-focused research meetings, engaged with various research collaborations, and been quite vocal about the need for change. In doing so, I have learned that the appetite is there, but that it is not so easy!
Operational and scientific barriers are hard to overcome when designing clinical trials that meet regulatory expectations, and contracting for partnerships between large organizations is wrapped up in complex legal discussions. There is certainly not a lack of interest in patient partnerships.
Without clear return on investment metrics, patient centricity risks becoming a checkbox exercise.
However, regulations are changing to allow for more long-term strategic involvement of patients through medicines development. For example, the UK revisions to the Clinical Trial Regulations, which come into effect in April 2026, aim to reduce unnecessary burden on participants (including long-overdue simplification of the informed consent process); make trials more flexible, inclusive, and responsive; and increase transparency and trust.11
Call to Action
Patient partnership offers a promising path forward. By involving patients throughout the development journey, the industry can create more relevant, effective, and trusted interventions. Now is the time to move from rhetoric to action—and build a future where treatments are truly designed for all of us.
References
- Everett Koop C. https://www.azquotes.com/author/8211-C_Everett_Koop. Accessed November 24, 2025.
- Davis J, Turnbull J, Reaney M. A patient experience data (PED) roadmap for patient-focused intervention development, determination and evaluation. In: Reaney M, ed. Using Patient Experience Data to Evaluate Medical Interventions. Generating, understanding and using patient experience data within and alongside clinical trials. IQVIA; 2023: 216-224.
- Oakley L. DrugsDisclosed.com launches unique ‘medication Trustpilot’ to amplify patient voices — as research reveals that 93% lack trust in pharma companies. https://www.businesswire.com/news/home/20200911005018/en/DrugsDisclosed.com-launches-unique-medication-Trustpilot-to-amplify-patient-voices-as-research-reveals-that-93-lack-trust-in-pharma-companies Published September 11, 2020. Accessed November 24, 2025.
- Reaney M, ed. Patient-centricity in the Biopharmaceutical Industry: Are We Nearly There Yet? A collection of perspectives from researchers, approvers and patients. IQVIA. https://www.iqvia.com/-/media/iqvia/pdfs/library/publications/2025/iqvia-patient-centricity-perspectives-book.pdf. Published July 30, 2025. Accessed April 13, 2026.
- Reaney M. A collection of essays on patient-centricity in intervention development [ISPOR Europe abstract PCR2]. Value Health. 2025;28(12):S547.
- Yanni AJ. A Bandana and a Bluebird: The Path to a Patient Centric Healthcare System. Amazon; 2024.
- Levitan B, Getz K, Eisenstein EL, et al. Assessing the financial value of patient engagement: A quantitative approach from CTTI’s patient groups and clinical trials project. Ther Innov Regul Sci. 2018;52(2):220-229.
- Nclusiv. Patient Engagement Statistics: Data That Proves Impact [blog]. https://nclusiv.co.uk/blog/f/patient-engagement-statistics-data-that-proves-impact Published June 20, 2025. Accessed March 2026.
- Cruz Rivera S, McMullan C, Jones L, et al. The impact of patient-reported outcome data from clinical trials: perspectives from international stakeholders. Journal Patient Rep Outcomes. 2020;4(1):1-14.
- Miller JD; Ruiz KM, Gagnon DD, et al. Assessing the impact of patient-reported outcomes and health economic benefit claims on the market value of oncologic pharmaceuticals [ISPOR Europe abstract PCN113]. Value Health. 2013;16(3):PA148.
- Alghabban A. The UK’s 2026 clinical trial regulations: A strategic analysis for global pharmaceutical sponsors. Ther Innov Regul Sci. 2026. doi:10.1007/s43441-026-00931-1

