Bridging Theory and Practice to Advance Patient-Centered Economic Evaluation of Health Technologies
Nan Qiao, PhD, ISPOR Patient-Centered Special Interest Group, Philadelphia, PA, USA; Andrew Briggs, DPhil, London School of Hygiene & Tropical Medicine, London, UK; Derick Mitchell, PhD, The Synergist, Brussels, Belgium

Economic evaluation is often a critical component of the health technology assessment (HTA) process. It attempts to provide a comprehensive analysis of the economic ramifications associated with integration of new health technologies into healthcare systems and inform reimbursement and pricing decision making. The goal is to allocate scarce resources to interventions that provide the most overall value to patients, healthcare systems, and society.
Patients are experts in living with their diseases and can offer valuable insights into the real-world impact of disease and health technologies. Integrating patient experiences into economic assessments can lead to decisions that more closely align with patient needs and values, thereby optimizing healthcare decision making and resource allocation.
This article will discuss the importance of patient-centered economic evaluation, how it is conducted, real-world examples, and courses of action to inspire broader adoption.
What Is Patient-Centered Economic Evaluation?
Defining a few key terms will provide a foundation for this discussion. Patient engagement in research is defined as “active, meaningful, and collaborative interaction between patients and researchers across all stages of the research process.”1 Patient experience data (PED) are “intended to provide information about patients’ experiences with a disease or condition,” including “the impact of such disease or condition, or a related therapy, on patients’ lives… [and] patient preferences with respect to treatment of such disease or condition.’’2 Patient-centered economic evaluation is conducted through the engagement of patients as research partners and the application of high-quality PED throughout HTA processes.
Patient-centered economic evaluation in HTA is often mistakenly viewed as incorporating patient out-of-pocket costs as inputs and/or estimating quality-adjusted life years as outputs. This view primarily reflects the perspectives of HTA bodies and researchers, and may not capture outcomes, values, and costs that matter to patients. The inclusion of those outcomes, values, and costs determines whether an evaluation is patient centered.
Patients are experts in living with their diseases and can offer valuable insights into the real-world impact of disease and health technologies.
Why Is It Important?
Patient-centered economic evaluation plays a crucial role in enhancing the scientific rigor of the economic model. Patient research partners and PED can provide valuable insights for establishing realistic assumptions grounded in actual patient experiences, creating a valid model structure that more accurately reflects disease progression and its impact, selecting the best available model inputs, and utilizing outcome measures that capture what is important to patients. Patient research partners can also help identify limitations within the economic model and highlight areas where the model may not adequately reflect their experiences or the complexities of their conditions and daily lives; this is essential for identifying potential biases and gaps in the model for decision makers’ consideration.
Incorporating the experiences and preferences of patients into the economic evaluation process ensures the evaluations better reflect the actual needs and priorities of patients, while fostering greater transparency and trust in the decision-making process. Ultimately, this alignment can result in more effective allocation of resources, improved health outcomes, and enhanced patient satisfaction within the healthcare system.
How to Conduct Patient-Centered Economic Evaluation
Recommendations from patient-centered research organizations and HTA bodies can guide the conduct of patient-centered economic evaluations. The Patient-Centered Outcomes Research Institute’s Foundational Expectations for Partnerships in Research,3 the Patient Focused Medicines Development’s Patient Engagement Quality Criteria,4 and the National Health Council’s (NHC) Rubric to Capture the Patient Voice5 provide fundamental principles for meaningful, effective, and sustainable patient engagement. Health Technology Assessment International and HTA bodies such as the National Institute for Health and Care Excellence and the Institute for Clinical and Economic Review have established high-level guidelines for patient engagement that may also be informative to patient-centered economic evaluations.6-8
The NHC’s Patient-Centered Value Model Rubric was developed to guide researchers in incorporating patient engagement into economic evaluations. It lists characteristics of meaningful patient engagement and considerations to enhance the patient-centeredness of economic evaluations with examples across 6 domains: patient partnership, transparency to patients, inclusiveness of patients, diversity of patients/populations, outcomes patients care about, and patient-centered data sources.9
Additionally, frameworks for engaging patients in other types of research have been leveraged by researchers to guide patient-centered economic evaluations. These include the 10-Step Framework for Continuous Patient Engagement in Comparative Effectiveness Research,10 the Conceptual Modeling Framework for Public Health Economic Modeling,11 and the Framework for Public Involvement in Mathematical and Economic Modelling.12
Beyond effective patient engagement, researchers should proactively seek high-quality PED to inform their work. PED encompasses patient experiences collected through a broad range of qualitative, quantitative, or mixed research methods. To be of value to multiple stakeholders, high-quality PED should be patient-engaged, meaningful, representative of diverse patient populations, and fit-for-purpose.13 Researchers must evaluate if published or newly collected PED accurately captures patient experiences, considering its patient-centricity, validity, and reliability, and transparently noting any limitations, such as lack of sample representativeness and associated constraints on generalizability, or potential bias introduced by missing data and nonresponse.14,15
Engagement of patients as research partners and high-quality PED are complementary: Engagement yields insights PED may miss, while PED can encompass broader patient populations. Together, they provide comprehensive insights that ensure evaluations are patient centered.16
Together, engagement of patients as research partners and high-quality patient experience data provide comprehensive insights that ensure economic evaluations are patient-centered.
Real-World Examples and Lessons Learned
We did not identify any publications on PED in economic evaluations as a component of HTA. Patient engagement remains uncommon in economic evaluations, although we identified 4 papers that describe engagement processes and their effects (Table).
- Mattingly et al17 assessed the cost-effectiveness of drug therapy for hepatitis C virus when considering non-health costs, such as patient/caregiver time and productivity, in addition to sustained virologic response.
- Al-Janabi et al18 worked with current or recent family caregivers to develop research methods for measuring and valuing the quality of life of family carers.
- Wilson et al19,20 engaged patients and caregivers in assessing the costs and benefits associated with chimeric antigen receptor T-cell (CAR-T) therapy for adults with acute lymphoblastic leukemia.
- Bunka et al21 included patient partners in the design and development of an economic simulation model of care pathways for major depressive disorder.
Patient engagement was operationalized using various methods, from consulting with individual patients19-21 to having a Lived Experience Advisory Panel of caregivers18 or a Stakeholder Advisory Board that included patients with other stakeholders.17 Advisory committees represent a widely recognized and effective method of stakeholder engagement. We recommend that researchers utilize a framework such as the NHC’s Rubric to Capture the Patient Voice5 to inform the governance and policies of these committees. We also recommend multiple patient partners representing diverse disease experiences. However, when resources such as funding, time, or personnel are limited, engaging 1 or 2 individual patient partners can be beneficial, although not optimal. Regardless of resource availability, researchers should have a solid understanding of the disease and patient life experiences by reviewing PED literature, in addition to consulting with patient partners.
Table. Patient Engagement and Impact Reported in Economic and Disease Modeling Publications
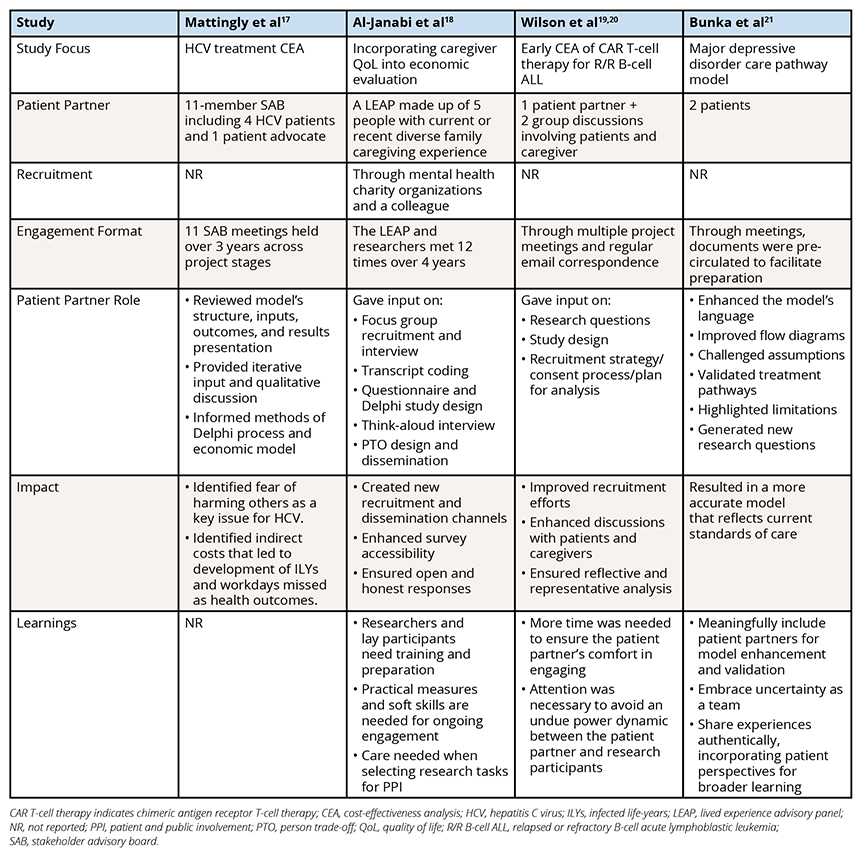
These studies provided a limited description of patient engagement processes. Patient partner recruitment was reported solely by Al-Janabi et al.18 Engagement formats were briefly described as meetings and/or emails,17-21 with Bunka et al noting that the precirculation of documents before meetings facilitated preparation.21 This lack of detail restricts our ability to fully assess the quality, variability, and effectiveness of engagement approaches and highlights the need for more comprehensive documentation and transparency.
Patient partners contributed to the studies by actively engaging in setting research questions, refining study designs, improving data collection and analysis, highlighting study limitations, creating new avenues for dissemination, and generating new research questions that could guide future investigations. Their contributions led to identification of important health outcomes, improved recruitment efforts, enhanced communication with patients and caregivers, facilitated a reflective and representative analysis, and resulted in more accurate models.17-21
Among the key engagement learnings from the studies, authors noted that effective collaborations require time and preparation, alongside practical measures and soft skills to ensure ongoing engagement.18-20 Wilson et al highlighted the potential for a power imbalance between the patient partner and patient research subjects, despite the fact that patient and public involvement helped equalize power between the academic team and the patient and caregiver participants.19,20 This underscores the importance of actively managing roles and expectations so that patient partners are empowered without inadvertently exerting influence over other patients’ contributions. We also emphasize the importance of recognizing and addressing power imbalances between researchers and patient partners as an important consideration in patient-centered research more generally.22,23
Al-Janabi et al suggested that researchers assign specific tasks for patient engagement.18 However, we contend that this approach can limit the scope of engagement and imply that patients are only capable of limited roles. Instead, patients should be engaged as equal research partners across all stages, fostering a genuinely collaborative environment. This can be operationalized by co-creating roles and responsibilities with patient partners, allowing flexibility and ongoing negotiation of contributions.
Discussion
As rising healthcare costs globally place unsustainable pressure on healthcare systems, many patient needs remain unmet. Patient-centered economic evaluation incorporates patient experiences to help ensure that healthcare resources are allocated to interventions addressing the most pressing needs of patients.
Despite examples of successful collaboration between researchers and patients, patient-centered economic evaluation in the context of HTA has not yet become commonplace. Few HTA guidelines require the incorporation of patient engagement and PED throughout the economic evaluation process. Guidelines’ emphasis on uniform inputs and standardized methods limits opportunities for meaningful patient engagement. In addition, researchers may underestimate the value of patient-centered economic evaluation and lack incentives to adopt this approach. Addressing these multifaceted barriers through multistakeholder approaches is essential to advancing patient-centered economic evaluation.
HTA bodies should collaborate with researchers and patients/patient groups to establish guidelines and standardized methods recommending that patient experiences be systematically considered in economic evaluations. They should also ensure that their decision-making
processes prioritize economic evaluations that better reflect patient perspectives. Researchers should receive targeted training to develop the skills needed for effectively engaging patients, including the ability to communicate complex concepts in plain language and gather meaningful patient input, supported by mentorship from patient engagement experts. Patients/patient groups should actively advocate for their inclusion in the economic evaluation process so that their lived experiences can inform more impactful research and healthcare decisions.
By fostering collaboration among HTA bodies, researchers, and patients/patient groups, the healthcare system can create a more inclusive environment for economic evaluations that are not only rigorous but also relevant and beneficial to the patients being served.
Disclosures: Nan Qiao is an employee of MSD. This work was written in her personal capacity, outside of her employment, and is unrelated to her role at MSD.
Acknowledgements: We would like to acknowledge Eleanor Perfetto, PhD; Anke-Peggy Holtorf, PhD, MBA; C. Daniel Mullins, PhD; and Bettina Ryll, MD, PhD, for their valuable inputs and contributions to this paper. We thank Jordana Foster, Managing Editor of VOS, for her thorough review and helpful comments on this manuscript.
References
- Harrington RL, Hanna ML, Oehrlein EM, et al. Defining patient engagement in research: Results of a systematic review and analysis. Report of the ISPOR Patient-Centered Special Interest Group. Value Health. 2020;23(6):677-688. doi: 10.1016/j. jval.2020.01.019.
- 21st Century Cures Act. HR 34, Pub L No. 114, Sec 3001(c). 114th Congress. https://www.congress.gov/114/plaws/publ255/PLAW-114publ255.pdf Published December 13, 2016. Accessed March 13, 2025.
- Patient-Centered Outcomes Research Institute. Foundational expectations for partnerships in research. https://www.pcori.org/engagement-research/engagement-resources/foundational-expectations. Accessed March 3, 2026.
- Patient Focused Medicines Development. Patient Engagement Quality Guidance. https://patientfocusedmedicine.org/peqg/patient-engagement-quality-guidance.pdf. Published May 2018. Accessed January 27, 2025.
- National Health Council. The National Health Council Rubric to Capture the Patient Voice: A Guide to Incorporating the Patient Voice into the Health Ecosystem. https://nationalhealthcouncil.org/wp-content/uploads/2019/12/NHC_Patient_Engagement_Rubric.pdf. Published June 2019. Accessed May 27, 2025.
- Health Technology Assessment International. Patient Involvement in HTA: Can We Learn From Past Experiences Across Europe? https://www.efpia.eu/media/nzqoxn0k/patient-involvement-in-hta-can-we-learn-from-past-experiences-across-europe.pdf. Published May 2024. Accessed January 27, 2025.
- National Institute for Health and Care Excellence. Patient and public involvement policy. https://www.nice.org.uk/about/nice-communities/nice-and-the-public/public-involvement/patient-and-public-involvement-policy. Last updated 2013. Accessed March 14, 2025.
- Institute for Clinical and Economic Review. A Guide to ICER’s Methods for Health Technology Assessment. https://icer.org/wp-content/uploads/2021/01/ICER_HTA_Guide_102720.pdf. Published October 27, 2020. Accessed March 14, 2025.
- National Health Council. The Patient Voice in Value: The National Health Council Patient-Centered Value Model Rubric. https://nationalhealthcouncil.org/wp-content/uploads/2019/12/Value-Rubric.pdf. Published March 2016. Accessed March 13, 2025.
- Mullins CD, Abdulhalim AM, Lavallee DC. Continuous patient engagement in comparative effectiveness research. JAMA. 2012;307(15):1587-1588. doi:10.1001/jama.2012.442.
- Squires H, Chilcott J, Akehurst R, Burr J, Kelly MP. A framework for developing the structure of public health economic models. Value Health. 2016;19(5):588-601. doi:10.1016/j.jval.2016.02.011.
- Staniszewska S, Hill EM, Grant R, et al. Developing a framework for public involvement in mathematical and economic modelling: Bringing new dynamism to vaccination policy recommendations [published correction appears in Patient. 2021 Jul;14(4):447]. Patient. 2021;14(4):435-445. doi:10.1007/s40271-020-00476-x.
- Schroeder K, Bertelsen N, Scott J, et al. Building from patient experiences to deliver patient-focused healthcare systems in collaboration with patients: A call to action. Ther Innov Regul Sci. 2022;56(5):848-858. doi:10.1007/s43441-022-00432-x.
- U.S. Food & Drug Administration. Patient-Focused Drug Development: Collecting Comprehensive and Representative Input. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/patient-focused-drug-development-collecting-comprehensive-and-representative-input. Published June 2018. Accessed March 6, 2026.
- European Medicines Agency. Patient experience data (PED) reflection paper. https://www.ema.europa.eu/en/patient-experience-data-ped-reflection-paper. Published September 29, 2025. Accessed March 6, 2026.
- Bertelsen N, Oehrlein E, Lewis B, et al. Patient engagement and patient experience data in regulatory review and health technology assessment: Where are we today? Ther Innov Regul Sci. 2025;59(4):737-752. doi:10.1007/s43441-025-00770-6.
- Mattingly TJ 2nd, Slejko JF, Onukwugha E, et al. Value in hepatitis C virus treatment: A patient-centered cost-effectiveness analysis. Pharmacoeconomics. 2020;38(2):233-242. doi:10.1007/s40273-019-00864-8.
- Al-Janabi H, Coles J, Copping J, et al. Patient and public involvement (ppi) in health economics methodology research: Reflections and recommendations. Patient. 2021;14(4):421-427. doi:10.1007/s40271-020-00445-4.
- Wilson M, Thavorn K, Hawrysh T, et al. Stakeholder engagement in economic evaluation: Protocol for using the nominal group technique to elicit patient, healthcare provider, and health system stakeholder input in the development of an early economic evaluation model of chimeric antigen receptor T-cell therapy. BMJ Open. 2021;11:e046707. doi:10.1136/bmjopen-2020-046707
- Wilson M, Thavorn K, Hawrysh T, et al. Engaging patients and caregivers in an early health economic evaluation: Discerning treatment value based on lived experience. Pharmacoeconomics. 2022;40(11):1119–1130. doi:10.1007/s40273-022-01180-4
- Bunka M, Ghanbarian S, Riches L, et al. Collaborating with patient partners to model clinical care pathways in major depressive disorder: The benefits of mixing evidence and lived experience. Pharmacoeconomics. 2022;40(10):971-977. doi:10.1007/s40273-022-01175-1.
- Richards DP, Bowden J, Gee P, et al. The ultimate power play in research - partnering with patients, partnering with power. Res Involv Engagem. 2025 Jun 17;11(1):65. doi: 10.1186/s40900-025-00745-9.
- Greenhalgh T, Hinton L, Finlay T, et al. Frameworks for supporting patient and public involvement in research: Systematic review and co-design pilot. Health Expect. 2019 Aug;22(4):785-801. doi: 10.1111/hex.12888.

