Addressing Barriers to Timely and Equitable Access in Oncology: The Case of Individualized Neoantigen Therapies
Federico Rodriguez-Cairoli, MD, Triangulate Health Ltd, Doncaster, UK; Meindert Boysen, PharmD, Independent HTA Expert, Arnhem, The Netherlands; Lieven Annemans PhD, Ghent University, Ghent, Belgium; David Tordrup MSc, Triangulate Health Ltd, Doncaster, UK; Michael Hartevelt, Merck & Co, Inc, Rahway, NJ, USA; Zine El Abidine Mosbahi, formerly MSD, Ottawa, Ontario, Canada

Nearly 6.2 million cancer deaths in the European Union and 1.3 million in the United Kingdom are estimated to have been prevented between 1989 and 2024, driven by reductions in risk factors, advancements in early detection, and significant progress in cancer management and therapy.1 Among the therapeutic innovations, precision medicine and advanced genetic and molecular technologies have demonstrated substantial improvements in survival and patient quality of life.2 However, as seen with the introduction of multi-indication medicines, healthcare systems must continuously adapt to ensure appropriate access to these therapies while managing their associated financial and logistical complexities.3-4
A key innovation in this landscape is the development of individualized neoantigen therapies (INTs).5 Designed to tailor treatment to each patient’s unique molecular tumor profile, INTs target specific neoantigens (proteins arising from tumor-specific mutations). This highly personalized approach could change future cancer care, offering potentially more effective and durable responses across a wide range of indications.6
Barriers to Access for Innovative Oncology Therapies
Novel oncology therapies often face barriers at launch due to financial constraints and uncertainties about their long-term clinical benefits. For instance, cell and gene therapies typically involve high upfront costs, yet the full scope of their effectiveness may take years to materialize.7 This delay may create uncertainty for payers and therefore complicate the assessment of their true value for healthcare systems, hindering patient access.8
The formal methodologies of cost-effectiveness analysis, often required early in health technology assessment (HTA) for reimbursement, add complexity to evaluating new therapy platforms. Often, short-term clinical evidence fails to capture the full value of these innovations, including their potential impact on the healthcare system and broader societal and economic benefits. There is a need for more comprehensive and forward-looking evaluation frameworks to fully recognize the value of novel platforms with the potential for significant scientific spillover effects that could help address a wide range of diseases in the future.
Unlike advanced therapies that target rare diseases, individualized neoantigen therapies have the potential to treat a larger patient population, particularly those in the earlier stages of cancer.
Unique Challenges for INTs
INTs introduce new considerations. Unlike other advanced therapies that target rarer diseases and smaller patient populations, INTs have the potential to treat a larger patient population, particularly those in the earlier stages of cancer, such as high-risk cutaneous melanoma.5 This wider applicability—while offering the potential to improve clinical outcomes broadly and reduce disease burden at a system level—may also carry broader budget implications for healthcare systems.
Importantly, the fact that INTs may find application in earlier-stage oncology indications introduces an additional layer of complexity for access and reimbursement. In such cases, novel therapies are often approved based on established clinical endpoints—such as disease-free survival (DFS) or recurrence-free survival (RFS). However, from the perspective of payers and HTA bodies, these endpoints may carry a degree of uncertainty regarding long-term outcomes, which can challenge reimbursement decision making and potentially delay patient access.9
Exploring Funding Mechanisms for Enhancing Access
Different funding mechanisms and payment models have been employed across a number of countries, particularly in Europe and the United Kingdom, to ensure timely access and reimbursement for innovative therapies in oncology. These approaches are not routinely applied for all submissions and remain the exception rather than the rule. A notable example is chimeric antigen receptor T-cell (CAR-T) therapies, which have been made accessible through financing approaches such as coverage with evidence development (CED) and performance-linked payment models.
Although CAR-T therapies are frequently referenced in discussions about innovative payment models, they differ from INTs in important ways. Due to their early oncology-stage indication and broader patient eligibility, INTs exhibit unique characteristics that may not be adequately addressed by existing payment models—highlighting the need to address the inherent limitations of current funding mechanisms and explore alternative approaches to ensure timely access.
The following provides a brief overview of these mechanisms, along with their potential for improvement. Additionally, Table 1 summarizes how current funding mechanisms address key considerations in ensuring timely access to INTs, along with their associated limitations.
Table 1. Applicability of current funding mechanisms for INTs
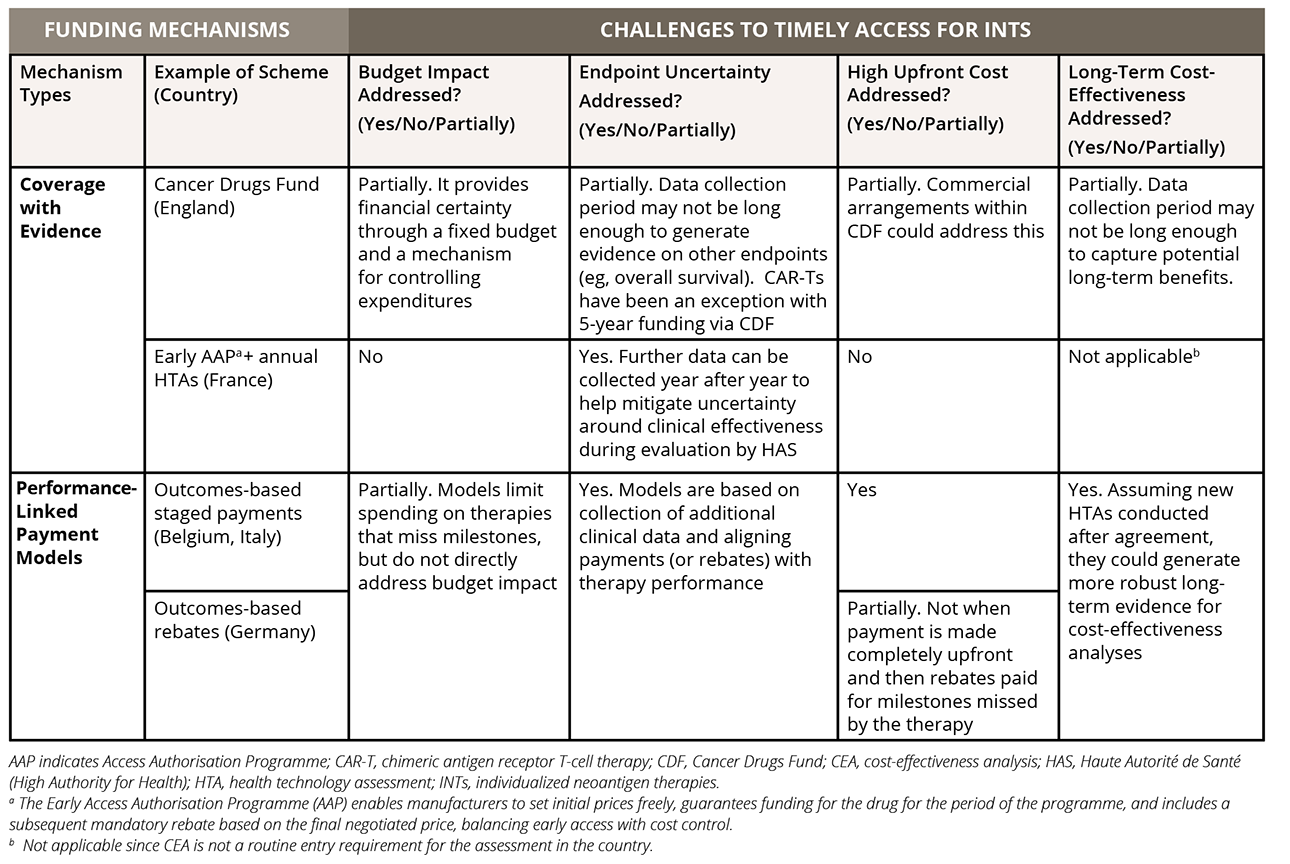
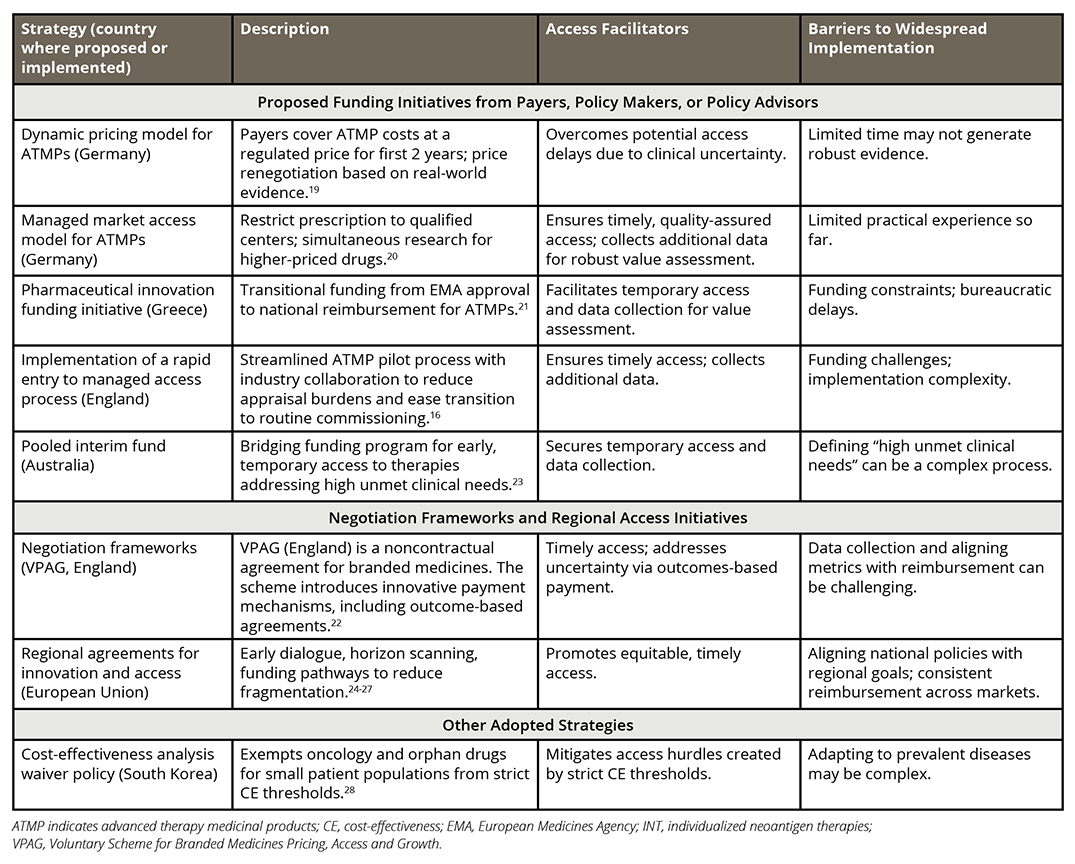
Table 2. Evaluation of alternative strategies for the application of INTs
Coverage With Evidence Development
France and England have employed CED models to facilitate access to CAR-T therapies for hematologic malignancies while additional evidence is collected to resolve assessment uncertainties.10,11
France implemented a CED approach with annual HTAs, alongside an early access model (formerly the Autorisation Temporaire d’Utilisation, now the Autorisation d’Accès Précoce). As part of this program, manufacturers must collect real-world safety and efficacy data on patients who receive early access to the therapy, and this information is used subsequently to complement clinical data from pivotal trials to inform HTA and reimbursement decisions. Clinical outcomes are also collected at specific intervals to inform HTA assessments; for therapies like tisagenlecleucel (Kymriah®) and axicabtagene ciloleucel (Yescarta®), these outcomes included survival rates, remission status, disease progression, and adverse events. This data collection was managed by the Lymphoma Academic Research Organization and reported quarterly by hospitals.10
In England, it was done through the Cancer Drugs Fund (CDF). Since its introduction the CDF has facilitated access to new cancer drugs while further evidence is gathered to address clinical uncertainties identified by the National Institute for Health and Care Excellence (NICE).11 For Kymriah, a key uncertainty for NICE involved overall survival and the proportion of patients who go on to have a stem cell transplant or require intravenous immunoglobulin after treatment. For Yescarta, a key uncertainty for NICE involved the choice of overall survival extrapolation in the cost-utility analysis, and the resulting uncertainty around the incremental cost-effectiveness ratio.12
Opportunities for Improvement. CED models continue to face significant implementation barriers. Although some health systems have initiated efforts to establish robust infrastructures for real-world data collection, a substantial number of jurisdictions lack the integrated data environments necessary to systematically capture health outcomes and resource utilization.13 Addressing these deficiencies through the development of interoperable health data platforms and the harmonization of national data governance frameworks will be critical to support rigorous evidence generation and informed decision making.
Many health systems lack the robust infrastructures needed for real-world evidence collection, which has limited the implementation of coverage with evidence-development funding models.
Performance-Linked Payment Models
Other countries have implemented performance-linked payment models when considering the integration of CAR-T therapies.12 In Germany, “outcomes-based rebates” have been employed, where the pharmaceutical company provides rebates to the payer based on individual outcomes, such as for patients who die within a (undisclosed) time frame following treatment with Kymriah. In Italy and Spain, “outcomes-based staged payments” have divided the total cost of the therapy into installments linked to patient outcomes, facilitated by national registry systems.12 Belgium has also adopted a pay-for-performance model, utilizing spread payments to align costs with therapeutic outcomes.14
With many stakeholders attracted to the concept of addressing clinical uncertainty by linking payments to specific performance metrics, there is growing consensus on the importance of adopting such innovative payment models. A recent report from the Organisation for Economic Co-operation and Development reveals that 16 of the 28 countries surveyed reported utilizing some form of performance-based managed entry agreement.15 Notably, Italy has successfully implemented outcomes-based reimbursement through the registry platform of the Italian Medicines Agency, enabling managed-entry agreements for various treatments, with 5 of the first 6 reimbursed Advanced Therapy Medicinal Products (ATMPs) in Italy utilizing such agreements to facilitate patient access.8
Another notable characteristic of these models, beyond addressing clinical uncertainty, is the option to facilitate payments in annuity installments, which can help mitigate cash flow and budget impact challenges. Although many stakeholders view the implementation of such payment structures as difficult due to national and EU-level accounting regulations serving as barriers, certain legal provisions in some countries could facilitate their adoption.16 These provisions have been seen in the context of both ATMP innovative payment models and broader financial reforms, such as the adaptation of budget cycles in Wales, where 3-year periods can be used to support long-term financial planning for innovative therapies,17 and France’s commitment to legislative changes allowing installment payments for advanced therapies.18
Opportunities for Improvement. While these models obviously better align payment and value, they also present challenges. Defining and measuring “performance” across various disease settings can be intricate, and establishing monitoring systems to generate the data needed for financial settlements may require considerable administrative effort.
The definition of “performance” in these models can vary significantly among countries and among payers, reflecting differences in regulatory frameworks and standards of evidence. This heterogeneity introduces considerable complexity in both the implementation of these models and the comparability of outcomes across healthcare systems. Establishing a harmonized framework for performance assessment may be critical for reducing variability, enabling cross-border learning, and facilitating the broader adoption of these models.
Finally, healthcare systems in which patients have the option of switching between different insurance providers must establish adequate procedures for risk allocation. This is heightened for those targeting non-orphan indications.
The European Union has been focused on equal access to innovation and competitiveness, with early dialogue mechanisms and funding pathways proposed to minimize fragmentation and complement national-level discussions.
Other Proposed Strategies
Beyond the funding mechanisms discussed above, several alternative strategies have been proposed or implemented across different jurisdictions to address barriers to timely access for innovative therapies.
In Germany, 2 distinct models have been proposed. One is a dynamic pricing model—proposed by Techniker Krankenkasse—whereby payers initially cover therapy costs at a regulated price for the first 2 years postlaunch, followed by price renegotiation based on real-world evidence and rebates if effectiveness is unsupported19; while this approach is designed to overcome access delays stemming from clinical uncertainty, the proposed time frame may be insufficient to generate robust evidence to support price negotiations. The second proposed model is a managed market access model, proposed by health insurance provider Barmer, which would restrict prescribing to specially qualified centers for higher-priced drugs with initially limited evidence, while simultaneously conducting application research20; this would facilitate quality-assured access and enable additional data collection for a more robust value assessment.
In Greece, a pharmaceutical innovation funding initiative has been proposed to provide transitional compensation for new treatments from EMA approval until inclusion in the national reimbursement list, focusing specifically on ATMPs.21 While this initiative would facilitate temporary access and additional data collection, it does not involve adjustments to the current value assessment framework, and challenges remain in regard to securing funding amid budget cuts and avoiding bureaucratic delays.
In England, beyond the CDF, the Association of British Pharmaceutical Industries has proposed a rapid entry to the managed access process, whereby NHSE and NICE would collaborate with industry using ATMPs as pilots; this plan would reduce appraisal burdens and streamline the transition to routine commissioning, ensuring timely patient access, although funding challenges and implementation complexity persist.16 Additionally, the 2024 Voluntary Scheme for Branded Medicines Pricing, Access and Growth—a noncontractual agreement between the UK government, the National Health Service in England, and the Association of British Pharmaceutical Industries—introduces innovative payment mechanisms including outcomes-based agreements that allow for pricing models facilitating immediate reimbursement; challenges remain around data collection and aligning performance metrics with reimbursement models.22
Australia’s HTA Review Reference Committee has recommended establishing a Bridging Funding Program providing early, temporary subsidized access to therapies addressing high unmet clinical needs, ensuring funding aligns with demonstrated benefits; the primary implementation challenge involves the complexity of defining what constitutes “high unmet clinical need” in practice.23
At the regional level, the European Union has been increasingly focused on equal access to innovation and competitiveness,24-26 with early dialogue mechanisms and funding pathways proposed to minimize fragmentation and complement national-level discussions. Aligning national policies with broader regional goals remains challenging; however, growing political momentum is evidenced by the adoption of more than half of the initiatives outlined in the Draghi report’s Competitiveness Compass within one year of its publication.27
Finally, South Korea has implemented a cost-effectiveness analysis waiver policy since 2015, exempting anticancer and orphan drugs targeting small patient populations with limited treatment options from strict cost-effectiveness thresholds, mitigating access hurdles, though adapting this model to more prevalent diseases may involve considerable complexity.28
Conclusion
The advent of innovative therapy platforms in oncology, such as INTs, presents a significant opportunity to explore solutions that ensure timely access for patients while maintaining the financial sustainability of healthcare systems.
Such solutions also require a framework for evidence creation to support regulatory approval and reimbursement, ensuring sufficient quality to limit uncertainty and support decision making and timely access for cancer patients.
Policy makers, payers, industry stakeholders, and patient groups must engage in constructive dialogue to address barriers and propose policy changes that ensure timely and equitable access to oncology advancements.
Disclosure statement
Mike Hartevelt is an employee of Merck Sharp & Dohme LLC (MSD), a subsidiary of Merck & Co, Inc, Rahway, NJ, USA. At the time of publication of this article, Zine El Abidine Mosbahi is no longer employed by MSD IDEA SPA; however, the work presented in this article was conducted while he was affiliated with the company. Federico Rodriguez-Cairoli and David Tordrup are employees of Triangulate Health Ltd. Triangulate Health Ltd received funding from MSD to conduct the research. Lieven Annemans and Meindert Boysen contributed to the article pro bono without receiving any honoraria.
Data availability
Data used in the paper are proprietary of MSD.
References
- Santucci C, Mignozzi S, Malvezzi M, et al. European cancer mortality predictions for the year 2024 with focus on colorectal cancer. Ann Oncol. 2024;35(3):308-316. doi:10.1016/j.annonc.2023.12.003
- Shifana AS, Adnan M, Gupta A, Ajazuddin, Jain P. A comprehensive review on novel pathways in cancer treatment: Clinical applications and future prospects. Curr Cancer Drug Targets. 2025;25(7):736-748. doi:10.2174/0115680096312603240709112520
- Lawlor R, Wilsdon T, Darquennes E, et al. Accelerating patient access to oncology medicines with multiple indications in Europe. J Mark Access Health Policy. 2021;9(1):1964791. doi:10.1080/20016689.2021.1964791
- Drummond M, Ciani O, Fornaro G, et al. How are health technology assessment bodies responding to the assessment challenges posed by cell and gene therapy? BMC Health Serv Res. 2023;23(1):484. doi:10.1186/s12913-023-09494-5
- Weber JS, Carlino MS, Khattak A, et al. Individualised neoantigen therapy mRNA-4157 (V940) plus pembrolizumab versus pembrolizumab monotherapy in resected melanoma (KEYNOTE-942): A randomised, phase 2b study. Lancet. 2024;403(10427):632-644. doi:10.1016/S0140-6736(23)02268-7
- Terai M, Sato T. Individualised neoantigen cancer vaccine therapy. Lancet. 2024 Feb 17;403(10427):590-591. doi:10.1016/S0140-6736(23)02463-7.
- Abuloha S, Niu S, Adirika D, Harvey BP, Svensson M. A review of the cost-effectiveness evidence for FDA-approved cell and gene therapies. Hum Gene Ther. 2024;35(11-12):365-373. doi:10.1089/hum.2023.186
- Rejon-Parrilla JC, Espin J, Garner S, Kniazkov S, Epstein D. Pricing and reimbursement mechanisms for advanced therapy medicinal products in 20 countries. Front Pharmacol. 2023;14:1199500. doi:10.3389/fphar.2023.1199500.
- Claessens Z, Fieuws S, Daems J, Barbier L, Huys I. Do European regulatory measures accelerate national market access in Belgium? A retrospective analysis of medicines centrally authorised between 2015 and 2020. BMJ Open. 2025;15(1):e091361. doi:10.1136/bmjopen-2024-091361.
- Jørgensen J, Kefalas P. The use of innovative payment mechanisms for gene therapies in Europe and the USA. Regen Med. 2021;16(4):405-422. doi:10.2217/rme-2020-0169
- NHS England. Cancer Drugs Fund. https://www.england.nhs.uk/cancer/cdf/ Accessed May 2025.
- Jørgensen J, Hanna E, Kefalas P. Outcomes-based reimbursement for gene therapies in practice: the experience of recently launched CAR-T cell therapies in major European countries. J Mark Access Health Policy. 2020;8(1):1715536. doi:10.1080/20016689.2020.1715536.
- Institute of Health Economics. Barriers and Enablers for Outcomes-based Agreements: An International Review. Edmonton (AB): Institute of Health Economics; 2023.
- Litvinova Y, Merkur S, Allin S, et al. Availability and financing of CAR-T cell therapies: A cross-country comparative analysis. Health Policy. 2024;149:105153. doi:10.1016/j.healthpol.2024.105153.
- Hofmarcher T, Berchet C, Dedet G. Access to oncology medicines in EU and OECD countries. https://www.oecd.org/en/publications/access-to-oncology-medicines-in-eu-and-oecd-countries_c263c014-en.html OECD Health Working Papers, No. 170. Published September 10, 2024. Accessed May 2025.
- The Association of the British Pharmaceutical Industry. Unlocking access to future ATMPs in the UK. https://www.abpi.org.uk/publications/unlocking-access-to-future-atmps-in-the-uk/ Published July 29, 2024. Accessed May 2025.
- Implementation of the NHS Finances (Wales) Act 2014. https://www.audit.wales/sites/default/files/nhs-finances-act-english-2017_6.pdf Published July 2017. Accessed May 2025.
- Ministère du Travail de la Santé et des Solidarités. Accord-cadre du 05/03/2021 entre le Comité économique des produits de santé et les entreprises du médicament (Leem). https://sante.gouv.fr/IMG/pdf/accord_cadre_21-24_signe.pdf Published March 5, 2021. Accessed May 2025.
- Caban A, Hammes F, Plesnila-Frank C, Toumi M. Is Germany going to open-up for outcomes-based agreements? [ISPOR Europe abstract PDG7]. Value Health. 2019;22(Suppl3):S598.
- Barmer. For a strong and stable healthcare system: Demands of BARMER for the 2025 federal election. https://www.barmer.de/resource/blob/1026282/26e4d744317a1292f139a5fee14977ab/gesundheitspolitische-positionen-der-barmer-2025-data.pdf Published December 2024. Accessed May 2025.
- Navlin Daily. Greece proposes innovation fund to improve access to innovative therapies. https://www.navlindaily.com/article/23172/greece-proposes-innovation-fund-to-improve-access-to-innovative-therapies Published September 23, 2024. Accessed May 2025.
- Health Technology Assessment Policy and Methods Review Reference Committee. Australian Government, Department of Health, Disability, and Aging. Health technology assessment policy and methods review. https://www.health.gov.au/our-work/hta-review Published September 10, 2024. Accessed May 2025.
- 2024 voluntary scheme for branded medicines pricing, access and growth: Summary of the heads of agreement. GOV.UK. https://www.gov.uk/government/publications/2024-voluntary-scheme-for-branded-medicines-pricing-access-and-growth-summary-of-the-heads-of-agreement Published November 20, 2023. Accessed May 2025.
- Reform of the EU pharmaceutical legislation. European Commission. https://health.ec.europa.eu/medicinal-products/reform-eu-pharmaceutical-legislation_en Last updated December 2025. Accessed March 2026.
- EU competitiveness – looking ahead. European Commission. https://commission.europa.eu/document/download/97e481fd-2dc3-412d-be4c-f152a8232961_en?filename=The%20future%20of%20European%20competitiveness%20_%20A%20competitiveness%20strategy%20for%20Europe.pdf. Published September 2024. Accessed May 2025.
- European Commission. Much more than a market – speed, security, solidarity. Empowering the Single Market to deliver a sustainable future and propensity for all EU citizens. https://www.consilium.europa.eu/media/ny3j24sm/much-more-than-a-market-report-by-enrico-letta.pdf Published April 2024. Accessed May 2025.
- European Commission. One year after the Draghi report: What has been achieved and what has changed. High-Level Conference. https://ec.europa.eu/commission/presscorner/api/files/attachment/881649/Factsheet%20-%20One%20year%20of%20The%20Draghi%20Report.pdf Published September 16, 2025. Accessed March 2026.
- Hong J, Bae EY, Lee HJ, Lee TJ, Clarke P. Values, challenges, and responses associated with high-priced potential cures: perspectives of diverse stakeholders in South Korea. Cost Eff Resour Alloc. 2024;22(1):20. doi:10.1186/s12962-024-00527-2.

